
For technical support call your local Bio-Rad office, or in the U.S., call 1-800-424-6723
Biotechnology Explorer
TM
Green Fluorescent Protein (GFP)
Purification Kit
Instruction Manual
Catalog #166-0005EDU
explorer.bio-rad.com
Duplication of any part of this document is permitted for classroom use only.
Please visit explorer.bio-rad.com to access our selection of language translations for
Biotechnology Explorer kit curriculum.

A Complete Teaching Guide
Developed over five years, Biotechnology Explorer kits and curricula have been written for
teachers, by teachers, and have been extensively field-tested in a broad range of classroom
settings from high school through the undergraduate level. Easy-to-use Biotechnology Explorer
kits are the perfect way to bring the excitement of biotechnology into the classroom. Each kit
contains an innovative step by step protocol, which makes them the perfect choice for both
experts and beginning teachers.
The curriculum contained within the manual for each kit makes our products unique. Each kit
contains its own unique curriculum package which is divided into a Teachers Guide and
Student Manual. The Teachers Guide is divided into three sections which help to insure that
the labs run smoothly. One section contains background information, lecture topics, and sug-
gested references which will enable each teacher, both experienced and newcomers to
biotechnology, to prepare and design lectures and lessons which can precede the actual labs.
This advance preparation will virtually insure that the labs run smoothly and that the students
understand the concepts behind each lab. There is a detailed section on the laboratory set up,
complete with simple procedures which contain graphic diagrams detailing the advance prepa-
ration for the labs. This makes the set up for each lab virtually foolproof. In addition, this sec-
tion contains time tables which will help you plan your schedule accordingly. Each lab can
be performed in a 50 minute time period, which should fit into most schedules.
Finally, we provide a detailed Teachers Answer Guide which contains answers to all of the
questions posed in the Student Manual. The teacher can use these answers as a guide when
reviewing or grading the questions presented in the Student section of the manual.
Each kit is designed to maximize student involvement in both the labs and the thought ques-
tions embedded in the manual. Student involvement in this process results in an increased
understanding of the scientific process and the value of proceeding into a task in an orga-
nized and logical fashion. Students who engage in the science curriculum found in the Bio-Rad
explorer kits develop a positive sense of their ability to understand the scientific method.
In order for your students to gain the most from this experiment they should know what a
gene is and understand the relationship between genes and proteins. For a more detailed
discussion of these and other basic molecular biology concepts and terms, refer to the review
provided in Appendix B.
We strive to continually improve our curriculum and products. Your input is extremely impor-
tant to us. Incorporation of your ideas, comments, critiques, and suggestions will enable the
explorer products to evolve into even better teaching aids.
You can find the catalog and curriculum on the Internet. Look up our home page at
explorer.bio-rad.com or call us at 1-800-424-6723.
Biotechnology Explorer Team
Bio-Rad Life Science Group
6000 James Watson Drive
Hercules, California 94547


Create context. Reinforce learning. Stay current.
New scientific discoveries and technologies
create more content for you to teach,
but not more time. Biotechnology
Explorer kits help you teach more
effectively by integrating multiple
core content subjects into a
single lab. Connect concepts
with techniques and put
them into context with
real-world scenarios.
GFP
Chromatography
Kit
Environmental
and Health
Science
• Pharmaceutical drug discovery
• Genetically modified
organisms (GMOs)
• GMOs in research, medicine,
nutrition, and bioremediation
• Microbiology
• Prokaryotic cell structure
and cell division
• Bacterial metabolism
• Selective growth media
• Use of chromatography to purify a
protein
• Interpretation of experimental
results
• Evaluation of hypotheses
• DNA > RNA > protein > trait
• Gene regulation and transcription
factors
• Protein structure (1
o
, 2
o
, 3
o
)
• Enzymes and catalysis
• Chemical properties of biological
molecules
• Hydrophobicity of proteins
• Chromatographic separation of
biomolecules
• Antibiotic selection and
resistance genes
• Selection mechanisms
• Adaptation to environment
Scientific
Inquiry
Chemistry
of Life
Genetics
Cell and
Molecular
Biology
Evolution


Table of Contents
Instructors Guide Page
Kit Inventory Check List Kit Components and Accessories ................................2
Safety Issues Working with
E. coli
, Disposal of
Biological Waste, and UV Lamps..................................3
Suggested Lesson Flow Planning Your Week ....................................................4
Workstation Check List Student and Instructor Lab Setups................................5
Implementation Timeline Advanced Prep and Student Lessons ..........................6
Lesson Highlights Detailed Instructors Guide ............................................9
Quick Guide Graphic Laboratory Protocol ......................................16
Teacher Answer Guide Answers to Student Review Questions ......................20
Student Manual
Green Fluorescent Protein (GFP) Purification ................................................................26
Lesson 1 Genetic Transformation Review: Finding the
Green Fluorescent Molecule ......................................27
Lesson 2 Inoculation Growing a Cell Culture..............................28
Lesson 3 Purification Phase 1 Bacterial Concentration
and Lysis ....................................................................32
Lesson 4 Purification Phase 2 Removing Bacterial Debris ........36
Lesson 5 Purification Phase 3 Protein Chromatography............39
Appendices
Appendix A Glossary of Terms ......................................................43
Appendix B Molecular Biology Concepts and Terminology ............47
Appendix C Sterile Technique........................................................52
Appendix D Gene Regulation and Genetic Selection ....................53

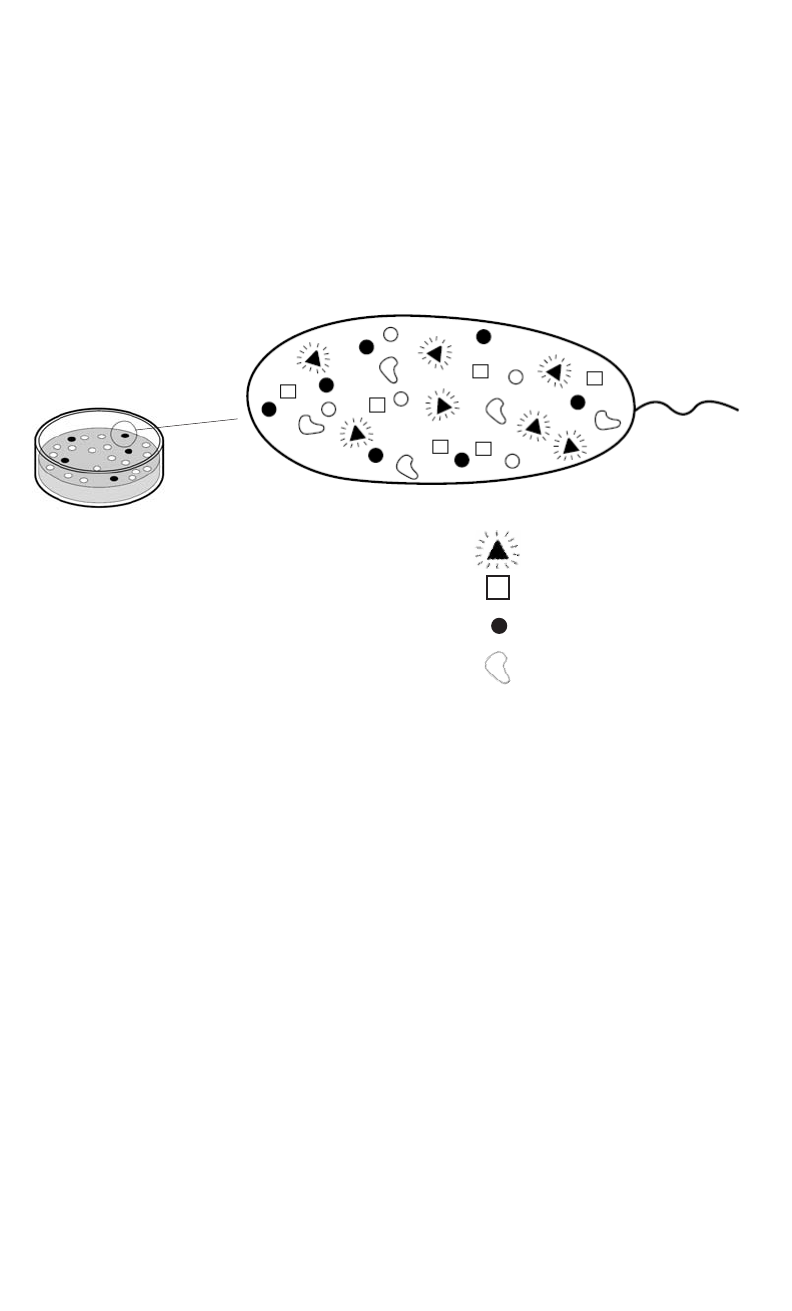
Is Finding a Needle in a Hay Stack Easier When it Glows?
This laboratory activity is designed to follow the pGLO
TM
Bacterial Transformation kit
(catalog #166-0003EDU). Students begin this activity with the bacteria they genetically trans-
formed using the plasmid, pGLO. Transformed bacteria which produce the genetically engi-
neered Green Fluorescent Protein (GFP) are removed from their agar plates and allowed to
multiply in liquid nutrient media. The bacterial cells are then broken open (lysed) to release the
Green Fluorescent Protein. GFP is subsequently purified from the contaminating bacterial
debris using the disposable chromatography columns provided in this kit. The unique fluo-
rescent properties of GFP allow the entire process to be observed using a long wavelength
UV lamp (i.e. pocket geology lamp).
One of the basic tools of modern biotechnology is DNA splicing, cutting DNA and
linking it to other DNA molecules. The basic concept behind DNA splicing is to remove a func-
tional DNA fragment—let’s say a gene—from one organism and combine it with the DNA of
another organism in order to make the protein that gene codes for. The desired result of gene
splicing is for the recipient organism to carry out the ge netic instructions provided by its newly
acquired gene. For example, certain plants can be given the genes for resistance to pests or
disease, and in a few cases to date, functional genes can be given to people with non-func-
tional or mutated genes, such as in a genetic disease like cystic fibrosis.
Genes can be cut out of human, animal, or plant DNA and placed inside bacteria.
For example, a healthy human gene for the hormone insulin can be put into bacteria. Under
the right conditions, these bacteria can make authentic human insulin. When allowed to mul-
tiply in gigantic vats (fermenters) these bacteria can be used to mass produce the human
insulin protein. This genetically engineered insulin is purified using protein chromatography and
used to treat patients with the genetic disease, diabetes, whose insulin genes do not function
normally.
A common problem in purifying genetically engineered “designer” proteins from trans-
formed bacteria is contamination by endogenous bacterial proteins. Chromatography is a
powerful method used in the biotechnology industry for separating and purifying proteins of
interest from bacterial proteins. Proteins purified in this manner can then be used, for exam-
ple, as medicines to treat human disease, or, for household agents such as natural enzymes
to make better laundry detergents.
The cloning and expression of the GFP gene (pGLO Bacterial Transformation kit), fol-
lowed by the purification of its protein in this kit, is completely analogous to the processes
used in the biotechnology industry to produce and purify proteins with commercial value. The
real-life source of the Green Fluorescent Protein gene is the bioluminescent jellyfish Aequoria
victoria. In this excercise, you may suggest a hypothetical scenario to your students in which
GFP has some special commercial value and its gene comes from a different natural source,
plant or animal. In either case, the principle is exactly the same, the gene codes for a Green
Fluorescent Protein.
“Bioengineered DNA was, weight for weight, the most valuable material in the world. A single
microscopic bacterium, too small to see with the human eye, but containing the gene for a heart
attack enzyme, streptokinase, or for “ice-minus” which prevented frost damage to crops, might
be worth 5 billion dollars to the right buyer.”
- Michael Crichton - Jurassic Park
1

Instructors Guide
Kit Inventory (✔) Checklist
This section lists the components provided in the GFP Purification Kit. It also lists required
accessories. Each kit contains sufficient materials to outfit eight student workstations. Use
this as a checklist to inventory your supplies before beginning the experiments.
Components Provided with the Kit Number/Kit (✔)
Ampicillin, lyophilized 1 vial ❏
Arabinose, lyophilized 1 vial ❏
LB broth capsule (to make 50 ml) 1 tablet ❏
Inoculation loops, packs of 10 loops 2 pk ❏
Pipets, sterile, individually wrapped 40 ❏
Microcentrifuge tubes, 2.0 ml, clear 30 ❏
Culture tubes, 15 ml, sterile (pack of 25) 1 pk ❏
Collection tubes, 5 ml, polystyrene 25 ❏
TE buffer (10 mM Tris, 1 mM EDTA, pH 8.0) 1 bottle ❏
Lysozyme, lyophilized 1 vial ❏
Binding buffer (4 M NH
4
SO
4
/TE, pH 8.0) 1 bottle ❏
Column equilibration buffer
(2 M NH
4
SO
4
/TE, pH 8.0) 1 bottle ❏
Column wash buffer (1.3 M NH
4
SO
4
/TE, pH 8.0) 1 bottle ❏
HIC chromatography columns 8 ❏
Column end caps 1 bag ❏
Accessories Not Included in the Kit Number/Class (✔)
Transformation plates
(pGLO Bacterial Transformation Kit
LB/amp and LB/amp/ara) 2 ❏
UV lamp, long wavelength*
(catalog #166-0500EDU) 1–4 ❏
Centrifuge (catalog #166-0602EDU) 1 ❏
Microwave oven 1 ❏
250 ml flask 1 ❏
100 ml graduated cylinder 1 ❏
Distilled water 100 ml ❏
Beaker of water for rinsing pipets 1 ❏
Marking pens 8 ❏
Refrigerator freezer 1 ❏
Microcentrifuge tube racks (catalog #166-0481EDU) 8 ❏
Optional Accessories
Shaking incubator, shaking water bath, tube roller
(catalog #166-0711EDU), or rocking platform
(catalog #166-0710EDU or 166-0709EDU) 1 ❏
Use of a rocker or shaker will speed bacterial
growth in liquid cultures but is not required.
Thermometer 1 ❏
2

Incubation oven (catalog #166-0501EDU) 1 ❏
Microcentrifuge (catalog #166-0602EDU) 1 ❏
Mini centrifuge (catalog #166-0603EDU) 1 ❏
Microtube centrifuge tube racks
(catalog #166-0481EDU) 8 ❏
Storage boxes (catalog #166-0482EDU) ❏
Refresh Kit Components
Catalog # Product Description
166-0015EDU Lyophilized components refill package, includes ampicillin,
arabinose, LB nutrient broth tablet, lysozyme
166-0406EDU Arabinose, 600 mg, lyophilized
166-0407EDU Ampicillin, 30 mg, lyophilized
166-0600EDU LB nutrient agar powder, 20 g, makes forty, 60 mm agar plates
166-0472EDU LB nutrient agar powder, 500 g, makes one thousand,
60 mm agar plates
166-0016EDU Chromatography buffers refill package, includes binding buffer,
column equilibration buffer, column wash buffer, elution buffer (TE)
166-0413EDU HIC chromatography columns and caps, 8 each
166-0471EDU Inoculation loops, 10 ml, sterile, 80
166-0474EDU Disposable plastic transfer pipets, sterile, 500
166-0480EDU Disposable plastic transfer pipets, nonsterile, 500
166-0476EDU Cell culture tubes, 17 x 100 mm, 14 ml, sterile, 25
223-9750EDU Clear polystyrene tubes, 13 x 100 mm, 9 ml, 1,000
166-0470EDU Petri dishes, 60 mm, sterile, 500
223-9430EDU EZ Micro
TM
Test Tubes, 2.0 ml, natural, 500
166-0479EDU Jellyfish foam floating racks, 8 racks with 12 microcentrifuge tube
wells
Safety Issues
The
Escherichia coli
bacteria HB101 K-12 strain contained in this kit is not a pathogenic organ-
ism like the
E. coli
strain O157 H7 that has sometimes been implicated in food poisoning.
HB101 K-12 has been genetically modified to prevent its growth unless grown on an enriched
medium. However, handling of the
E. coli
K-12 strain requires the use of standard
Microbiological Practices. These practices include, but are not limited to, the following. Work
surfaces are decontaminated once a day and after any spill of viable material. All contaminated
liquid or solid wastes are decontaminated before disposal. All persons must wash their hands:
(i) after they handle material containing bacteria, and (ii) before exiting the laboratory. All pro-
cedures are performed carefully to minimize the creation of aerosols. Mechanical pipeting
devices are used, mouth pipetting is prohibited; eating, drinking, smoking, and applying cos-
metics are not permitted in the work area; wearing protective eyewear and gloves is strong-
ly recommended.
If an autoclave is not available, all solutions and components (loops and pipets) that have
come in contact with bacteria can be placed in a fresh 10% bleach solution for at least
20 min for sterilization. A shallow pan of this solution should be placed at every lab station. No
matter what you choose, all used loops and pipets should be collected for sterilization. Sterilize
petri dishes by covering the agar with 10% bleach solution. Let the plate stand for
1 hr or more, and then pour excess plate liquid down the drain. Once sterilized, the agar
plates can be double bagged and treated as normal trash. Safety glasses are recommended
when using bleach solutions.
3

4
Ampicillin may cause allergic reactions or irritation to the eyes, respiratory system, and skin.
In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
Wear suitable protective clothing. Ampicillin is a member of the penicillin family of antibiotics.
Those with allergies to penicillin or to any other member of the penicillin family of antibiotics
should avoid contact with ampicillin.
Please obtain the Material Safety Data Sheets (MSDS) available from Bio-Rad by calling 800-
424-6723 in the United States, or see www.bio-rad.com for further information on reagents in
this kit. Please consult your local environmental health and safety regulations for proper dis-
posal.
*UV lamp
Ultraviolet radiation can cause damage to eyes and skin. Short-wave UV is more damaging
than long-wave UV light. The Bio-Rad UV lamp recommended for this module is long-wave.
If possible, use UV-rated safety glasses or goggles.
Suggested Lesson Flow
There are five lessons in the GFP Purification kit curriculum, including four active student lab-
oratory sessions. All lessons are designed to be carried out in consecutive 50 min periods. For
continuity, the five lessons can be conveniently started on a Monday and completed on a
Friday. Step by step protocols employed in the student labs are detailed in the Student Manual.
Lesson 1 Introduction to Purification. Lecture and discussion.
Lesson 2 Picking Colonies and Inoculating Cell Cultures
Lesson 3 Purification Phase 1—Bacterial Concentration and Lysis
Lesson 4 Purification Phase 2—Removing Bacterial Debris
Lesson 5 Purification Phase 3—Protein Chromatography
Advance Preparation Instructors Overview
This section outlines the recommended schedule for advance preparation on the part of the
instructor. A detailed advance preparation guide begins on page 6.
Prep Activity When Time required
Read manual Immediately 1 hour
Prepare liquid culture media Before lesson 2 15 minutes
Set up workstations Day of each lesson 5 minutes/day

Workstations Daily Inventory Check (✔) List
Student Workstations
Materials and supplies that should be present at each student workstation site prior to begin-
ning each lab activity are listed below. The components provided in this kit are sufficient to out-
fit 8 complete student workstations.
Instructors (Common) Workstation
Materials, supplies, and equipment that should be present at a common location that can be
accessed by all students during each lab activity are also listed below. It is up to the discre-
tion of the teacher as to whether students should access common buffer solutions/equipment,
or whether the teacher should aliquot solutions and operate equipment.
5
Lesson 2
Student workstations Number/team
Transformation plates
from pGLO Bacterial
Transformation kit
(LB/amp/ara and LB/amp) 2
Inoculation loops 2
Culture tubes containing
2 ml growth media 2
Marking pen 1
Microcentrifuge tube rack 1–4
Instructors workstation
Shaking incubator, shaking
water bath, tube roller, or
rocking platform (optional) 1
UV light 1–4
Lesson 3
Student workstations
Microcentrifuge tubes 1
Pipets 1
Microcentrifuge tube rack 1
Marking pen 1
Beaker of water for
rinsing pipets 1
Instructors workstation
TE buffer 1 bottle
Lysozyme (rehydrated) 1 vial
Centrifuge 1
UV light 1–4
Lesson 4
Student workstations Number/team
Microcentrifuge tubes 1
Pipets 1
Microcentrifuge tube rack 1
Marking pen 1
Beaker of water for
rinsing pipets 1
HIC chromatography column 1
Column end cap 1
Instructors workstation
Binding buffer 1 bottle
Equilibration buffer 1 bottle
Centrifuge 1
UV light 1–4
Lesson 5
Student workstations
Collection tubes 3
Pipets 1
Microcentrifuge tube rack 1
Marking pen 1
Beaker of water for
rinsing pipets 1
HIC chromatography
column 1
Column end cap 1
Instructors workstation
Wash buffer 1 bottle
Equilibration buffer 1 bottle
TE buffer 1 bottle
UV light 1–4

Instructors Advance Preparation for Labs
This section describes preparation to be performed in advance by the instructor for the active lab
sessions in Lessons 2 through 5.
Lesson 2 Inoculation—Growing a Cell Culture
Lesson 3 Purification Phase 1—Bacterial Concentration and Lysis
Lesson 4 Purification Phase 2—Removing Bacterial Debris
Lesson 5 Purification Phase 3—Protein Chromatography
For students to begin Lesson 2 they will need the two transformation plates (LB/amp/ara and
LB/amp) from the pGLO Bacterial Transformation kit. To avoid contamination, bacterial cells
from these plates should be used within 1 week following the transformation activity. At the
completion of the transformation activity, store the transformation plates in a refrigerator to keep
the cells fresh.
Lesson 2 Inoculation—Growing a Cell Culture
Advance Preparation
Objectives Prepare liquid culture media (6–8 below)
Set up student and instructors workstations (Page 5)
Set up rocking table or shaking incubator or incubator oven
Time Required 30 minutes
Note: Observe sterile technique while preparing the following materials. See
Appendix C.
Prepare Ampicillin and Arabinose Solutions
Ampicillin and arabinose are shipped dry in small vials. After being rehydrated, both are added
to the liquid growth media. Ampicillin is an antibiotic which inhibits growth of bacterial con-
taminants which may be introduced from the environment. Arabinose is a sugar which induces
the overexpression of the Green Fluorescent Protein in cloned cells. Refer to Appendix A and
D for more details on the functions of these two components.
Using a sterile pipet, add 3 ml of TE buffer directly to the vial containing the ampicillin. Using
another sterile pipet, add 3 ml of TE buffer to rehydrate the arabinose. Mix the vials and gen-
tly swirl or vortex to aid in rehydration.
Note: Rehydrate ampicillin and arabinose the day you prepare the liquid growth
medium. Arabinose requires 10 min to dissolve—be patient.
6
TE buffer
1 ml
ArabinoseAmpicillin

7
Prepare Liquid Nutrient Media
In Lesson 2, each student workstation will require two culture tubes containing 2 ml of liquid
nutrient media. These will be used to grow bacterial cultures. To prepare the liquid media,
add 55 ml of distilled water to a 250 ml Erlenmeyer flask and heat to boiling in a microwave.
Then, add the single LB capsule to the flask. Let the tablet soak in the hot water for 20 min;
this will aid in dissolving. Heat the flask again to boiling in the microwave. Swirl the flask to dis-
solve the tablet. Repeat heating and swirling several times until the entire tablet is dissolved,
but be careful to allow the flask to cool a little before swirling so that the hot medium does
not boil over onto your hand.
When the entire tablet is dissolved, allow the LB to cool so that the outside of the flask is com-
fortable to hold, or below 50°C. While the media is cooling, get the ampicillin and arabinose
solutions that were prepared in step 1 above. When the media has cooled, use a new pipet
and transfer 0.5 ml of arabinose and 0.5 ml of ampicillin into the flask. Swirl the flask to mix
the components. You can now discard the residual arabinose and ampicillin solutions.
Aliquot Liquid Culture Media
Using a new pipet as above, aliquot 2 ml of the liquid media into 16 culture tubes. (This can
be accomplished by transferring the media in two 1 ml aliquots from a sterile pipet.) Store the
culture tubes in a refrigerator until the day of use. Nine extra culture tubes are provided. You
may wish to fill these tubes with media and use them for demonstrations or for backup liquid
cultures for those student teams that do not have successful growth.
For Lesson 2, each student workstation will need two culture tubes, each containing 2 ml of
liquid culture media. In this lesson, students inoculate their 2 ml cultures with transformed bac-
teria from the pGLO Bacterial Transformation kit.
Microwave
to boiling
Add water
Swirl
Add tablet
Microwave
to boiling
Add
arabinose
and
ampicillin
Swirl

Note: Ideally, cultures should be shaken at 40 –200 rpm at 32°C for 24 hr. Shaking provides
oxygen to the dividing cells promoting growth, and the more shaking, the better. This
is traditionally achieved using a shaking incubator or water bath. Alternatively, culture
tubes can be capped tightly and spun for 24 hr using a tube roller inside an incubation
oven set to 32°C. If such a devices are not available, cell cultures may be shaken
manually for 30 sec and incubated at 32°C for 24 hr. Simply have the students shake
the capped culture tubes for 30 sec, as they would a can of spray paint. The more
vigorous the manual shaking, the more growth, which results in greater production of
GFP. After shaking, lay the tubes on their sides in the incubator oven at 32°C for 1–2
days. When the tubes are oriented horizontally, more surface area of the culture media
is exposed to the air in the tube, allowing more oxygen to diffuse into the cells. Shake
periodically during the 2 day incubation.
If no incubator is available, cells will grow less well but sufficiently at room
temperature after 2 days. However, cells must be shaken either using a tube roller or
a rocker during this incubation. Stationary cultures are not recommended for room
temperature growth. Following the appropriate incubation period, cell cultures
inoculated with white and green transformed colonies should appear bright green under
ultraviolet light.
Lesson 3 Purification Phase 1
Bacterial Concentration and Lysis
Advance Preparation
Objective Set up workstations
Rehydrate lysozyme
Set up centrifuge
Time required 10 minutes
Rehydrate the vial of lyophilized lysozyme with 1 ml of TE buffer using a new pipet. Mix
gently to aid in the resuspension. Keep the vial of lysozyme on ice or in a refrigerator until
use.
Lesson 4 Purification Phase 2
Removing Bacterial Debris
Objective Set up workstations (the only preparation needed)
Set up centrifuge
Time required 10 minutes
Lesson 5 Purification Phase 3
Protein Chromatography
Objective Set up workstations (the only preparation needed)
Time required 10 minutes
Should the columns dry out, they can be easily rehydrated by doing the following: Pour the
resin into a beaker or other container and add water to make a slurry. Then pour the slurry
back into the column. Drain out the water and equilabrate the column in the binding buffer.
8

Lesson Points to Highlight
This section describes steps in the experimental protocols which may be technically chal-
lenging or which are extremely important to the overall outcome and understanding of the
experiments. Instructors should alert their students attention to these points, and when pos-
sible, demonstrate the technique before the students attempt the procedure.
The Student Manual contains the detailed descriptions and drawings of all laboratory steps and
the techniques employed in Lessons 2–5. Refer to it for questions about the experimental
protocols used in lab.
Use of the Pipet
Before beginning the laboratory sessions, point out the graduations on the pipet to the stu-
dents. Both the 250 µl and 1 ml marks will be used as units of measurement throughout
Lessons 2 through 5.
Lesson 1 Genetic Transformation Review
Growth Media
The liquid and solid agar media are referred to as LB (named after Luria-Bertani) broth and
are made from an extract of yeast and an enzymatic digest of meat byproducts which provides
a mixture of carbohydrates, amino acids, nucleotides, salts, and vitamins, all of which are
nutrients for bacterial growth. Agar, which is derived from seaweed, melts when heated and
cools to form a solid gel (very analogous to Jell-O), and functions to provide a solid support
on which to culture bacteria.
9
250 µl
100 µl
500 µl
750 µl
1 ml

Antibiotic Selection
The pGLO plasmid which contains the GFP gene also contains the gene for beta-lactamase,
a protein that provides bacteria with resistance to the antibiotic, ampicillin. The beta-lacta-
mase protein is produced and secreted by bacteria which contain the plasmid. The secreted
beta-lactamase inactivates ampicillin, allowing only transformed cells to grow in its presence.
Only transformed bacteria which contain the pGLO plasmid, and produce beta-lactamase
can survive in the presence of ampicillin. (See schematic below.)
Gene Regulation
One of the main instructional highlights of the pGLO Transformation Kit was the concept of
gene regulation. In this kit, the bacteria which were transformed with the pGLO plasmid were
plated onto LB/amp and LB/amp/ara agar plates. Expression of the GFP gene is under the reg-
ulatory control of the arabinose promoter. Thus, when the bacteria were plated onto LB agar
containing arabinose (LB/amp/ara), GFP was expressed and the colonies appeared bright
green. Conversely, when the bacteria were plated onto LB agar that did not contain arabinose
(LB/amp), the gene was turned off and the colonies appeared white. (See Appendix D for more
details.)
The concept of gene regulation can be further expanded upon in this kit. If a white colony
(which contains the GFP gene, but is not expressing GFP in the absence of arabinose) is
seeded into LB/amp/ara liquid media, the arabinose present in the media will be taken up by
the bacteria, which subsequently turns on the GFP gene. Thus the students will be seeding
a white colony which has the dormant GFP gene into arabinose containing media. Arabinose
turns on the dormant gene, resulting in a fluorescent green liquid culture. In addition, the stu-
dent will be seeding a green fluorescent colony into LB/amp/ara liquid culture. In this case
the gene will remain "on" and the liquid culture will also fluoresce bright green.
Bacterial proteins
Arabinose promotor protein
Green fluorescent protein
Beta-lactamase protein
Bacterial
chromosomal
DNA
10
Genetically engineered plasmid used to insert new genes
into bacteria.
"GFP" Gene which codes for the Green Fluorescent Protein.
"
bla
" Gene which codes for beta-lactamase, a protein which
gives bacteria resistance to the antibiotic, ampicillin.
"
araC
" Gene which codes for AraC, a protein which regulates
production of the Green Fluorescent Protein.
Lesson 2 Inoculation—Growing a Cell Culture
Isolation of Single Bacterial Colonies
In this activity, students will pick one white colony from their LB/amp plates and one green
colony from their LB/amp/ara plates for propagation in parallel liquid cultures.
Cloning describes the isolation and propagation of a single gene. Because a single bacterial
colony originates from a single bacterium, all the individual bacteria in the colony are genet-
ically identical and they are called clones. When the students pick colonies (or clones) of both
green and white bacteria from their agar plates, single isolated colonies are chosen for trans-
fer to the culture media. Single isolated colonies which are separated from other colonies on
the agar by at least 1–2 mm are generally not contaminated with other bacteria. The schemat-
ic below illustrates the genes expressed in fluorescent green colonies.
The instructor may want to demonstrate how to scoop a single, well-isolated colony from the
agar using an inoculation loop. Again, it is very important that proper sterile technique is fol-
lowed during the picking of colonies and subsequent dissociation of the colonies into the cul-
ture tubes. Remind students to keep their plates covered and their tubes capped wherever
possible.
Overnight Liquid Cultures
The GFP gene requires a 32°C (or lower) incubation temperature for optimal protein folding
and fluorescence. If a 32°C incubator is unavailable, the bacteria can be cultured by shaking
at room temperature, but this will require a 48 hr, rather than a 24 hr, culture period.
In this lesson, liquid bacterial cultures will yield sufficient growth and production of protein sim-
ply by incubating overnight at 32°C. However, vigorous shaking of the liquid culture delivers
more oxygen to the dividing cells allowing them to grow faster and produce more GFP. For
this reason, we strongly recommend that a shaking incubator, shaking water bath, or a tube
roller used inside an incubation oven is used. If a shaking incubator is not available, cell cultures
can be shaken manually for 30 sec then incubated 1–2 days at 32°C. Simply shake the capped
culture tubes vigorously for 30 seconds – like a can of spray paint. Then place the tubes on
their sides in the incubator oven for 1–2 days. Placing the tubes horizontally in the oven max-
imizes the culture surface area and oxygen exchange. Removing the tubes and shaking them
periodically during the incubation period will enhance cell growth further.
11
Green fluorescent protein (GFP)
Antibiotic resistance protein
Bacterial proteins
GFP regulator protein
Green fluorescent
colonies (•)
Transformed bacterial cell

The more constant and more vigorous the shaking, the more growth and the more production
of GFP. In either case, following incubation, the liquid culture should fluoresce bright green
upon exposure to UV light.
Culture Condition Days Required
32°C—shaking or rolling 1 day
32°C—no shaking 1–2 days*
Room Temperature—shaking or rolling 2 days
Room Temperature—no shaking Not recommended
* Periodically shake by hand and lay tubes horizontally in incubator.
Lesson 3 Purification Phase 1
Bacterial Concentration and Lysis
Centrifugation
Centrifugation is a technique used to separate molecules on the basis of size by high speed
spinning (somewhat analogous to the spin cycle in a washing machine where the clothes
become compacted against the walls of the washer). In this lab session, the heavier bacteri-
al cells will be separated from the liquid growth media by a single centrifugation step.
Centrifugation results in a "pellet" of bacteria found at the bottom of the tube, and a liquid
"supernatant" that resides above the pellet. The collection and concentration of bacteria is a
first step in the isolation of GFP from the bacteria that were grown in the liquid media in Lesson
2.
Using a disposable pipet, students transfer the 2 ml of culture from the culture tube into a
2 ml microcentrifuge tube—in one gentle squirt. They will then spin the bacterial pellet down
to the bottom of the microcentrifuge tube and pour off (discard) the liquid supernatant above
the pellet. The pellet will fluoresce bright green upon exposure to UV light because the green
protein is being expressed within the bacteria. At this point, this short discussion of a "pellet"
and "supernatant" may be helpful.
12
Transformed bacterial cell
Supernatant
Bacterial
pellet
Green fluorescent protein (GFP)
Antibiotic resistance protein
Bacterial proteins
GFP regulator protein

Resuspension of the Bacterial Pellet
In this step of the protocol, it is essential that the entire bacterial pellet is resuspended in TE
buffer. The students should add 250 µl of TE buffer to the pellet of bacteria, and then resuspend
the pellets by carefully, but rapidly, pipetting the cells up and down with the 250 µl volume of
buffer. (If vortexes are available, gentle vortexing will also aid in resuspension.) The volume
in the tube can be visually inspected for large chunks of unresuspended bacteria. If chunks
are observed, the students should continue to pipet up and down to fully resuspend the bac-
teria.
Lysozyme
Lysozyme is an enzyme that functions to degrade (or lyse) the bacterial cell wall, by cleaving
polysaccharide (sugar) residues in the cell walls. The subsequent freeze-thaw step used in this
lesson aids in the complete disruption of the wall and internal membrane. Complete disrup-
tion or "lysis" releases soluble components, including GFP. Lysozyme is naturally found in
human tears, acting as a bactericidal agent to help prevent bacterial eye infections. Lysozyme
gets its name from its natural ability to "lyse" bacteria.
Lesson 4 Purification Phase 2
Removing Bacterial Debris
This final centrifugation step serves to separate the large particles of lysed bacteria (such as
the cell membrane and walls) from the much smaller proteins, including GFP. The larger bac-
terial debris are pelleted in the bottom of the microcentrifuge tube, while the smaller proteins
remain in the supernatant. At this stage, the supernatant will fluoresce bright green upon
exposure to UV light. Students should then, very carefully, remove the supernatant from the
pellet and place it into a new 2 ml microcentrifuge tube using a clean 1 ml pipet. This should
be done immediately to prevent pellet debris from leaching and contaminating the super-
natant and potentially clogging the chromatography column in the purification procedure of les-
son 5.
Lesson 5 Purification Phase 3
Protein Chromatography
Chromatography is a powerful technique for separating proteins in a complex mixture. Bacteria
contain thousands of endogenous proteins from which GFP must be separated. In chro-
matography, a cylinder, or column is densely filled with a "bed" of microscopic beads. These
beads form a matrix through which proteins must pass before being collected. The matrix the
students will employ in this case has an "affinity" for the molecule of interest (GFP), but not
for the other bacterial proteins in the mixture. GFP "sticks" to the column, allowing it to be
separated from the bacterial contaminants.
13
+ Lysozyme
Freezing +
centrifugation
Bacterial pellet
(cell wall and debris)
Resuspended
bacterial pellet

Hydrophobic ("water hating") substances do not mix well with water. When they are dropped
into salty water they tend to stick together. Some of the amino acids that make up proteins are
very hydrophobic. In salt water, these parts of a protein tend to stick tightly to other hydropho-
bic substances. High salt levels cause the three-dimensional structure of the protein to actu-
ally change so that the hydrophobic regions of the protein are more exposed on the surface
of the protein and the hydrophilic ("water-loving") regions are more shielded.
A chromatography column which is packed with hydrophobic beads is called a hydrophobic
interaction matrix. When the sample is loaded onto the matrix in salt water, the hydrophobic
proteins in the sample will stick to the beads in the column. The more hydrophobic they are
the more tightly they will stick. When the salt is removed, the three-dimensional structure of
the protein changes again so that the hydrophobic regions of the protein now move to the
interior of the protein and the hydrophilic ("water-loving") regions move to the exterior. The
result is that the hydrophobic proteins no longer stick to the beads and drip out the bottom of
the column, separated from the other proteins.
Hydrophobic Interaction Chromatography (HIC)
In lessons 4 and 5, the soluble GFP in the supernatant is purified using hydrophobic interac-
tion chromatography (HIC). GFP has several stretches of hydrophobic amino acids, which
results in the total protein being very hydrophobic. When the supernatant, rich in GFP, is
passed over a HIC column in a highly salty buffer (Binding Buffer), the hydrophobic regions
of the GFP stick to the HIC beads. Other proteins which are less hydrophobic (or more
hydrophilic) pass right through the column. This single procedure allows the purification of
GFP from a complex mixture of bacterial proteins.
Loading the GFP supernatant onto the chromatography column
When students load the GFP supernatant onto their columns, it is very important that they
do not disturb the upper surface of the column bed when performing the chromatography
procedure. The column matrix should have a relatively flat upper surface. A slightly uneven
column bed will not drastically affect the procedure. However, subsequent steps of loading,
washing, and eluting should minimize disrupting the column such that beads "fluff up" into
the buffer. When loading the GFP supernatant onto the column, the pipet tip should be insert-
ed into the column and should rest against the side of the column. The supernatant should be
slowly expelled from the pipet, down the walls of the column. When the supernatant has com-
pletely entered the column, a green ring of fluorescence should be visible at the top of the
bed when viewed with the UV light.
There are four different buffers which are used in the HIC procedure. Each buffer should be
slowly pipetd down the side of the column to minimize disturbance to the column resins.
Equilibration buffer
A medium salt buffer (2 M (NH
4
)
2
SO
4
) which is used to "equilibrate" or "prime" the chro-
matography column for the binding of GFP.
Binding buffer
An equal volume of high salt binding buffer (4 M (NH
4
)
2
SO
4
) is added to the bacterial lysate. The
end result is that the supernatant containing GFP has the same salt concentration as the equi-
librated column. When in a high salt solution, the hydrophobic regions of proteins are more
exposed and are able to interact with and bind the hydrophobic regions of the column.
Wash buffer
A medium salt wash buffer (1.3 M (NH
4
)
2
SO
4
) is used to wash weakly associated proteins from
the column; proteins which are strongly hydrophobic (GFP) remain bound to the column. When
the wash buffer is applied to the column, care should again be taken to minimize disturbance
14

15
of the column resin. Slow, steady pipetting down the side of the column is most effective. At this
point "a ring" of GFP should begin to penetrate the upper surface of the matrix (~ 1–2 mm into
the bed).
Elution buffer
A low salt buffer (TE buffer; 10 mM Tris/EDTA) is used to wash GFP from the column. In low
salt buffers (which have a higher concentration of water molecules), the conformation of GFP
changes so that the hydrophilic residues of GFP are more exposed to the surface, causing the
GFP to have a higher affinity for the buffer than for the column, thereby allowing the GFP to
wash off the column. The 750 ml of TE (elution) buffer is applied to the column gently, as
described above. The TE buffer will disrupt the hydrophobic interactions between the GFP and
the column bed, causing GFP to let go and "elute" from the column. The GFP should pass
down the column as a bright green fluorescent ring. This is easily observed using the UV
light. If the column bed was disturbed in any of the preceding steps, the GFP will not elute as
a distinct ring, but will elute with a more irregular, distorted shape. However, elution should
still occur at this step. If successful, collection tube 3 should fluoresce bright green.
Storage of tubes
All of the collection tubes and their constituents can be parafilmed or covered and stored for
approximately 1–2 weeks in the refrigerator.
Important hints for successful chromatography
1. Place the column gently into the collection tubes. Jamming the column tightly into the
collection tubes will create an air tight seal and the sample will not flow through. You
can create a “paper crutch” by folding a small piece of paper, about the size of a match
stick, and wedging it between the column and the collection tube. This crutch makes it
impossible for an air tight seal to form, and insures that the column will flow.
2. The flow rate of the column can be increased in the elution step by placing the top cap
tightly back onto the column. This creates air pressure which pushes on the column
bed, causing the sample to flow faster.
3. The columns are designed to drip slowly. The entire chromatography procedure should
take 20 to 30 min. It is important not to remove the column more than needed from col-
lection tube to collection tube, as motion can cause major disturbance to the column
bed.
Binding
Wash
Elution

GFP Purification—Quick Guide
16
Incubate at 32°C 24 hr
or
48 hr at room
temperature
➔
Lesson 2 Inoculation
Growing Cell Cultures
1. Remove the transformation plates
from the incubator and examine using
the UV light. Identify several green
colonies that are not touching other
colonies on the LB/amp/ara plate.
Identify several white colonies on the
LB/amp plate.
2. Obtain two culture tubes containing
the growth media LB/amp/ara. Label
one "+" and one "-". Using a sterile
loop, lightly touch the loop to a green
colony and immerse it in the "+" tube.
Using a new sterile loop, repeat for a
white colony and immerse it in the "-"
tube (it is very important to pick only a
single colony). Spin the loop between
your index finger and thumb to dis-
perse the entire colony.
3. Cap the tubes and place them in the
shaking incubator, shaking water
bath, tube roller, or rocker and culture
for 24 hr at 32°C or 2 days at room
temperature.
or
Cap the tubes and shake vigorously
by hand. Place in the incubator hori-
zontally at 32°C for 24–48 hr. Remove
and shake by hand periodically when
possible.
LB/ampLB/amp/ara
LB/amp
LB/amp/ara
+
-

17
Lesson 3 Purification Phase 1
Bacterial Concentration
1. Label one microcentrifuge tube "+" with
your name and class period. Remove
your liquid cultures from the shaker and
observe with the UV light. Note any
color differences between the two cul-
tures. Using a new pipet, transfer 2 ml
of "+" liquid culture into the “+” micro-
centrifuge tube. Spin the microcentrifuge
tube for 5 minutes in the centrifuge at
maximum speed. The pipet used in this
step can be repeatedly rinsed in a
beaker of water and used for all follow-
ing steps of this laboratory period.
2. Pour out the supernatant and observe
the pellet under UV light.
3. Using a rinsed pipet, add 250 µl of TE
buffer to the tube. Resuspend the pellet
thoroughly by rapidly pipetting up and
down several times.
4. Using a rinsed pipet, add 1 drop of
lysozyme to the resuspended bacterial
pellet to initiate enzymatic digestion of
the bacterial cell wall. Mix the contents
gently by flicking the tube. Observe the
tube under the UV light.
5. Place the microcentrifuge tube in the
freezer until the next laboratory period.
The freezing causes the bacteria to rup-
ture completely.
250 µl TE
2 ml
1 drop lysozyme
++
+
Freezer
➔
➔

18
Lesson 4 Purification Phase 2
Bacterial Lysis
1. Remove the microcentrifuge tube
from the freezer and thaw using hand
warmth. Place the tube in the cen-
trifuge and pellet the insoluble bacte-
rial debris by spinning for 10 minutes
at maximum speed.
2. While your tube is spinning, prepare
the chromatography column. Remove
the cap and snap off the bottom from
the prefilled HIC column. Allow all of
the liquid buffer to drain from the col-
umn (~3–5 minutes).
3. Prepare the column by adding 2 ml
of Equilibration Buffer to the top of the
column. This is done by adding two 1
ml aliquots with a rinsed pipet. Drain
the buffer to the 1 ml mark on the col-
umn. Cap the top and bottom and
store the column at room temperature
until the next laboratory period.
4. After the 10 minute spin, immediate-
ly remove your tube from the cen-
trifuge. Examine the tube with the UV
light. Using a new pipet, transfer 250
µl of the "+" supernatant into a new
microcentrifuge tube labeled "+".
Again, rinse the pipet well for the rest
of the steps of this lab period.
5. Using a well rinsed pipet, transfer 250
µl of binding buffer to the "+" super-
natant. Place the tube in the refriger-
ator until the next laboratory period.
Equilibration buffer (2 ml)
250 µl
250 µl
1 ml
Centrifuge
Thaw
+
+
➔

19
Lesson 5 Purification Phase 3
Protein Chromatography
1. Label 3 collection tubes 1–3 and place
the tubes in the foam rack or in a rack
supplied in your laboratory. Remove the
caps from the top and bottom of the col-
umn and place the column in collection
tube 1. When the last of the buffer has
reached the surface of the HIC matrix
proceed to the next step below.
2. Using a new pipet, carefully and gently
load 250 µl of the “+” supernatant onto
the top of the column. Hold the pipet tip
against the side of the column wall, just
above the upper surface of the matrix
and let the supernatant drip down the
side of the column wall. Examine the col-
umn using a UV light. Note your obser-
vations. After it stops dripping transfer
the column to collection tube 2.
3. Using the rinsed pipet, add 250 µl of
wash buffer and let the entire volume
flow into the column. Examine the col-
umn using the UV light. Note your obser-
vations. After the column stops dripping,
transfer it to tube 3.
4. Using the rinsed pipet, add 750 µl of TE
Buffer and let the entire volume flow into
the column. Examine the column using
the UV light. Note your observations.
5. Examine all three collection tubes and
note any differences in color between
the tubes. Parafilm or plastic wrap the
tubes and place in the refrigerator until
the next laboratory period.
1
1
2
Wash buffer (250 µl)
(250 µl) + supernatant
TE buffer (750 µl)
3
3
2
1
+

Teacher Answer Guide
Lesson 1 Review Questions
1. Proteins
a. What is a protein ?
A macromolecule which consists of chains of amino acids.
b. List three examples of proteins found in your body.
Antibodies, digestive enzymes, hair proteins, hormones, and hemoglobin are all
examples of proteins.
c. Explain the relationship between genes and proteins.
Genes contain the genetic code which determine the amino acid composition of
a protein. There is a unique gene for each protein within all of the cells of the
body.
2. Using your own words, define or describe cloning.
The duplication and propagation of a cell or organism.
3. Describe how bacterial cells in a "library" are different from the cells of a single colony.
A library of bacterial cells contains a diverse mixture of bacterial types which
contains a diverse mixture of genes. A single colony of bacteria originates from
an individual clone which only contains a single gene.
4. Describe how you might recover the cancer-curing protein from the bacterial cells.
One can isolate a single fluorescent green colony of bacteria and grow large
amounts of the bacteria in a liquid growth media. Bacteria in liquid media can be
concentrated by centrifugation. After the bacterial cells are lysed to release the
cancer curing protein, the protein can be isolated by passage through a chro-
matography column which has an affinity for the cancer-curing protein.
20

Lesson 2 Review Questions
1. What is a bacterial colony?
A bacterial colony is a large group or cluster of bacterial cells that originated
from a single, clonal cell.
2. How are these items helpful in this cloning experiment?
a. ultraviolet (UV) light—The green protein fluoresces, and is thus visible,
when exposed to the UV light.
b. incubator—The incubator provides a warm temperature which enables the
bacteria to grow.
c. shaking incubator—A shaking incubator provides a warm temperature and
provides aeration which oxygenates the bacterial cultures. Increased oxy-
gen accelerates the growth rate of bacteria.
3. Can you predict what would happen if you took one of the green colonies from the
LB/amp/ara plate and streaked it onto an LB/amp plate? Conversely, what would hap-
pen if you took a white colony from the LB/amp plate and streaked it onto an
LB/amp/ara plate? Explain your answer.
If a green colony was streaked onto an LB/amp plate, the resulting colonies
would be white. This plate does not contain arabinose which is needed to induce
expression of the GFP gene and generate green fluorescent colonies. If a white
colony was streaked onto an LB/amp/ara plate, the resulting colonies would be
green. This plate contains arabinose which induces expression of the GFP gene
and generates green fluorescent colonies.
21

Lesson 3 Review Questions
1. You have used a bacterium to propagate a gene that produces a green fluorescent pro-
tein. Identify the function of these items you used in Lesson 3.
a. Centrifuge—Functions to pellet the bacteria and separate the bacteria from
the growth media.
b. Lysozyme—Functions to enzymatically digest the bacterial cell well, which
in turn weakens the cell wall so that it will rupture upon freezing.
c. Freezer—Functions to freeze the bacteria which causes the cytoplasm to
expand, which completely ruptures the weakened cell wall.
2. Can you explain why both liquid cultures fluoresce green?
The green colony seeded into the (+) tube fluoresces green because the arabi-
nose in the liquid culture media continues to induce expression of the GFP gene,
which results in a green culture. The white colony seeded into the (+) tube fluo-
resces green because the arabinose in the liquid culture media turns on expres-
sion of the GFP gene which was previously off on the LB/amp plate (which lacks
arabinose), which results in a green culture.
3. Why did you discard the supernatant in this part of the protein purification procedure?
The supernatant contains the bacterial growth media and does not contain the
desired GFP.
4. Why did you discard the white liquid from the "-" tube but keep the green one?
The white culture of bacteria does not contain the GFP and is not needed for the
subsequent purification step.
5. Can you explain why the bacterial cells' outer cell wall ruptures when the cells are
frozen? What happens to an unopened soft drink when it freezes?
When a bacterial cell freezes, the volume of cytoplasm expands. The expansion
puts pressure on the weakened cell wall, which then ruptures from the pressure.
6. What was the purpose of rupturing or lysing the bacteria?
The bacteria need to be ruptured in order to release the GFP, which can then be
purified using column chromatography.
22

Lesson 4 Review Questions
1. What color was the pellet in this step of the experiment? What color was the super-
natant? What does this tell you?
The pellet should be a whitish or pale green color. The supernatant should fluo-
resce bright green. The fluorescent green color of the supernatant indicates that
the green protein was released from the bacteria and remained in the super-
natant. The much lighter color of the bacterial pellet suggests that the GFP was
released from the bacteria upon lysis.
2. Why did you discard the pellet in this part of the protein purification procedure?
The pellet contains unwanted bacterial debris—bacterial cell walls, membranes,
and chromosomal DNA. The pellet contains little, if any, GFP and can be
discarded.
3. Briefly describe hydrophobic interaction chromatography and identify its purpose in this
lab.
Protein chromatography is a technique which can be used to separate or purify
proteins from other molecules. This lab used hydrophobic interaction chro-
matography to purify GFP based upon its hydrophobic properties.
23

Lesson 5 Review Questions
1. List your predictions and observations for the sample and what happens to the sample
when the following buffers are added to the HIC column.
Observations
Under UV Light
Collection Tube Number Prediction (column and collection tube)
Tube 1 GFP should stick to the GFP resides as a band at
Sample in binding buffer column the top of the column
Tube 2 GFP should stick to the GFP remains as a broad
Sample in wash buffer column band on top of the column
Tube 3 GFP should elute from GFP travels down
Sample with elution buffer the column column as a ring and
elutes into tube 3
2. Using the data table above, compare how your predictions matched up with your obser-
vations for each buffer.
a. Binding Buffer—GFP binds to the top of the chromatography column.
b. Wash Buffer—GFP remained bound to the top of the column.
c. Elution Buffer—GFP is eluted from the column.
3. Based on your results, explain the roles or functions of these buffers. Hint: how does
the name of the buffer relate to its function?
a. Equilibration Buffer—This buffer prepares the column for the application of
GFP. Equilibration buffer raises the salt concentration of the column to
match that of the bacterial GFP lysate.
b. Binding Buffer—This buffer raises the salt concentration of GFP which
causes a conformational change in GFP, exposing the hydrophobic
regions.
c. Wash Buffer—Wash buffer functions to wash away less hydrophobic, con-
taminating proteins from the column.
d. TE (elution) Buffer—This buffer functions to remove GFP from the column.
4. Which buffers have the highest salt content and which have the least? How can you
tell?
Binding buffer >>Equilibration buffer>>Wash buffer>>Elution buffer
Binding buffer has the highest concentration of salt because it is needed to raise
the salt concentration of the GFP lysate. The hydrophobic patches of proteins are
exposed in high salt buffer. TE elution buffer has the lowest salt concentration
because it causes GFP to elute from the column. The hydrophobic patches of
proteins re-orient to the interior, and the hydrophilic regions are exposed in low
salt buffer.
24

5. Were you successful in isolating and purifying GFP from the cloned bacterial cells?
Identify the evidence you have to support your answer.
If tube 3 fluoresces green, the student was successful in purifying GFP. If GFP is
not present in tube 3, examine the column—application of an incorrect buffer
would prevent the elution. Alternatively, if the student did not start with a bright
green culture, then tube 3 will not be extremely bright.
25

Green Fluorescent Protein (GFP)
Purification
Student Manual
"Bioengineered DNA was, weight for weight, the most valuable material in the world. A
single microscopic bacterium, too small to see with the human eye, but containing
the gene for a heart attack enzyme, streptokinase, or for "ice-minus" which pre-
vented frost damage to crops, might be worth 5 billion dollars to the right buyer."
Michael Crichton - Jurassic Park
Contents
Lesson 1 Genetic Transformation Review—Finding the Green Fluorescent Molecule
Lesson 2 Inoculation—Growing a Cell Culture
Lesson 3 Purification Phase 1—Bacterial Concentration and Lysis
Lesson 4 Purification Phase 2—Removing Bacterial Debris
Lesson 5 Purification Phase 3—Protein Chromatography
26

Lesson 1 Finding the Green Fluorescent Molecule
Genetic Transformation Review
With the pGLO Bacterial Transformation kit, you performed a genetic transformation of
E. coli
bacterial cells. The results of this procedure were colonies of cells that fluoresced when
exposed to ultraviolet light. This is not a normal phenotype (characteristic) for
E.coli
. You
were then asked to figure out a way to determine which molecule was becoming fluorescent
under UV light. After determining that the pGLO plasmid DNA was not responsible for the flu-
orescence under the UV light, you concluded that it was not the plasmid DNA that was fluo-
rescing in response to the ultraviolet light within the cells. This then led to the next hypothesis
that if it is not the DNA fluorescing when exposed to the UV light, then it must be a protein that
the new DNA produces within the cells.
1. Proteins.
a. What is a protein?
b. List three examples of proteins found in your body.
c. Explain the relationship between genes and proteins.
2. Using your own words, describe cloning.
3. Describe how the bacterial cloned cells on your LB/amp plate differ from the cells on
your LB/amp/ara plate. Can you design an experiment to show that both plates of
cloned cells behave similarly and do contain the same DNA?
4. Describe how you might recover the cancer-curing protein from the bacterial cells.
27

Laboratory Procedure for Lesson 2
Picking Colonies and Growing a Cell Culture
Examine your two transformation plates under the ultraviolet (UV) lamp. On the LB/amp plate
pick out a single colony of bacteria that is well separated from all the other colonies on the
plate. Use a magic marker to circle it on the bottom of the plate. Do the same for a single
green colony on the LB/amp/ara plate. Theoretically both white and green colonies were
transformed with the pGLO plasmid? How can you tell?
Both colonies should contain the gene for the Green Fluorescent Protein. To find out, you
will place each of the two different bacterial colonies (clones) into two different culure tubes
and let them grow and multiply overnight.
Your Task
In this lab, you will pick one white colony from your LB/amp plate and one green colony from
your LB/amp/ara plate for propagation in separate liquid cultures. Since it is hypothesized
that the cells contain the Green Fluorescent Protein, and it is this protein we want to produce
and purify, your first consideration might involve thinking of how to produce a large number
of cells that produce GFP.
You will be provided with two tubes of liquid nutrient broth into which you will place cloned cells
that have been transformed with the pGLO plasmid.
Workstation Daily Inventory Check (✔) List
Your Workstation. Materials and supplies that should be present at your student worksta-
tion site prior to beginning this lab activity are listed below.
Instructors (Common) Workstation. Materials, supplies, and equipment that should be
present at a common location that can be accessed by your group during each lab activity are
also listed below.
Your workstation Number (✔)
Transformation plates from pGLO Bacterial
Transformation kit (LB/amp/ara and LB/amp) 2 ❏
Inoculation loops 2 ❏
Culture tubes, containing 2 ml growth media 2 ❏
Marking pen 1 ❏
Test tube holder 1 ❏
Instructors workstation
Shaking incubator, shaking water bath,
tube roller or rocking platform (optional) 1 ❏
UV light 1 ❏
28
Laboratory Procedure for Lesson 2
1. Examine your LB/amp and LB/amp/ara plates from the transformation lab. First use
normal room lighting, then use an ultraviolet light in a darkened area of your laboratory.
Note your observations.
To prevent damage to your skin or eyes, avoid exposure to the UV light. Never look
directly into the UV lamp. Wear safety glasses whenever possible.
2. Identify several green colonies that are not touching other colonies on the LB/amp/ara
plate. Turn the plate over and circle several of these green colonies. On the other
LB/amp plate identify and circle several white colonies that are also well isolated from
other colonies on the plate.
3. Obtain two culture tubes containing 2 ml of nutrient growth media and label one tube
"+" and one tube "–". Using a sterile inoculation loop, lightly touch the "loop" end to a
circled single green colony and scoop up the cells without grabbing big chunks of agar.
Immerse the loop in the "+" tube. Spin the loop between your index finger and thumb to
disperse the entire colony. Using a new sterile loop, repeat for a single white colony and
immerse it in the "–" tube. It is very important to pick cells from a single bacterial colony.
29
LB/ampLB/amp/ara
LB/ampLB/amp/ara
+
-

4. Cap your tubes and place them in the shaking incubator, shaking water bath, tube roller
or rocker. Let the tubes incubate for 24 hr at 32°C or for 2 days at room temperature. If
a shaker is not available, shake your two tubes vigorously, like you would shake a can
of spray paint, for about 30 sec. Then place them in an incubator oven for 24 hr. Lay
the tubes down horizontally in the incubator. (If a rocking table or tube roller is avail-
able, but no incubator, tape the tubes to the platform or insert in tube roller and let them
rock or spin at maximum speed for 24 hr at 32°C or at room temperature for 48 hr. We
do not recommend room temperature incubation without rocking or shaking.)
Culture Condition Days Required
32°C—shaking or rolling 1 day
32°C—no shaking 1–2 days*
Room temperature—shaking or rolling 2 days
Room temperature—no shaking Not recommended
* Periodically shake by hand and lay tubes horizontally in incubator.
30
Cap the tubes.
Incubate at 32° C overnight
or
48 hr at
room temperature.
➔

Lesson 2 Name________________
Review Questions
1. What is a bacterial colony?
2. Why did you pick one green colony and one white colony from your agar plate(s)? Why
do you think you picked one of each color? What could this prove?
3. How are these items helpful in this cloning experiment?
a. ultraviolet (UV) light -
b. incubator -
c. shaking incubator -
4. Explain how placing cloned cells in nutrient broth to multiply relates to your overall goal
of purifying the fluorescent protein.
31

Lesson 3
Purification Phase 1
Bacterial Concentration and Lysis
So far you have mass produced living cultures of two cloned bacterium. Both contain the
gene which produces the green fluorescent protein. Now it is time to extract the green protein
from its bacterial host. Since it is the bacterial cells that contain the green protein, we first
need to think about how to collect a large number of these bacterial cells.
A good way to concentrate a large number of cells is to place a tube containing the liquid cell
culture into a centrifuge and spin it. As you spin the cell culture, where would you expect the
cells to concentrate, in the liquid portion or at the bottom of the tube in a pellet?
Workstations Check (✔) List
Your Workstation. Make sure the correct materials listed below are present at your work-
station prior to beginning this lab experiment.
Instructors (Common) Workstation. Materials that should be present at a common loca-
tion to be accessed by your group are also listed below.
Your workstation Number (✔)
Microcentrifuge tubes 1 ❏
Pipets 1 ❏
Microcentrifuge tube rack 1 ❏
Marking pen 1 ❏
Beaker of water for rinsing pipets 1 ❏
Instructors workstation
TE buffer 1 bottle ❏
Lysozyme (rehydrated) 1 vial ❏
Centrifuge 1 ❏
UV light 1–4 ❏
32

Laboratory Procedure for Lesson 3
1. Using a marker, label one new microcentrifuge tube with your name and period.
2. Remove your two liquid cultures from the shaker or incubator and observe them in nor-
mal room lighting and then with the UV light. Note any color differences that you
observe. Using a clean pipet, transfer the entire contents of the (+) liquid culture into the
2 ml microcentrifuge tube also labeled (+), then cap it. You may now set aside your (–)
culture for disposal.
3. Spin the (+) microcentrifuge tube for 5 minutes in the centrifuge at maximum speed. Be
sure to balance the tubes in the machine. If you do not know how to balance the tubes,
do not operate the centrifuge.
4. After the bacterial liquid culture has been centrifuged, open the tube and slowly pour off
the liquid supernatant above the pellet. After the supernatant has been discarded, there
should be a large bacterial pellet remaining in the tube.
5. Observe the pellet under UV light. Note your observations.
6. Using a new pipet, add 250 µl of TE buffer to each tube. Resuspend the bacterial pellet
thoroughly by rapidly pipetting up and down several times with the pipet.
33
1 ml
++
+
250 µl TE
+

7. Using a rinsed pipet, add 1 drop of lysozyme to the resuspended bacterial pellet. Cap
and mix the contents by flicking the tube with your index finger. The lysozyme will start
digesting the bacterial cell wall. Observe the tube under the UV light. Place the micro-
centrifuge tube in the freezer until the next laboratory period. The freezing will cause the
bacteria to explode and rupture completely.
34
1 drop lysozyme

Lesson 3 Name _______________
Review Questions
1. You have used a bacterium to propagate a gene that produces a green fluorescent pro-
tein. Identify the function of these items you need in Lesson 3.
a. Centrifuge -
b. Lysozyme -
c. Freezer -
2. Can you explain why both liquid cultures fluoresce green?
3. Why did you discard the supernatant in this part of the protein purification procedure?
4. Can you explain why the bacterial cells’ outer membrane ruptures when the cells are
frozen. What happens to an unopened soft drink when it freezes?
5. What was the purpose of rupturing or lysing the bacteria?
35

Lesson 4
Purification Phase 2
Removing Bacterial Debris
The bacterial lysate that you generated in the last lab contains a mixture of GFP and endoge-
nous bacterial proteins. Your goal is to separate and purify GFP from these other contami-
nating bacterial proteins. Proteins are long chains of amino acids, some of which are very
hydrophobic or "water-hating". GFP has many patches of hydrophobic amino acids, which
collectively make the entire protein hydrophobic. Moreover, GFP is much more hydrophobic
than most of the other bacterial proteins. We can take advantage of the hydrophobic proper-
ties of GFP to purify it from the other, less hydrophobic (more hydrophilic or "water-loving") bac-
terial proteins.
Chromatography is a powerful method for separating proteins and other molecules in complex
mixtures and is commonly used in biotechnology to purify genetically engineered proteins. In
chromatography, a column is filled with microscopic sperical beads. A mixture of proteins in
a solution passes through the column by moving downward through the spaces between the
beads.
You will be using a column filled with beads that have been made very hydrophobic—the
exact technique is called hydrophobic interaction chromatography (HIC). When the lysate is
applied to the column, the hydrophobic proteins that are applied to the column in a high salt
buffer will stick to the beads while all other proteins in the mixture will pass through. When the
salt is decreased, the hydrophobic proteins will no longer stick to the beads and will drip out
the bottom of the column in a purified form.
Workstations Check (✔) List
Student Workstations. Make sure the materials listed below are present at your worksta-
tion prior to beginning this lab experiment.
Instructors (Common) Workstation. Materials that should be present at a common loca-
tion to be accessed by your group are also listed below.
Student workstation items Quantity (✔)
Microcentrifuge tubes 1 ❏
Pipets 1 ❏
Microcentrifuge tube rack 1 ❏
Marking pen 1 ❏
Beaker of water for rinsing pipets 1 ❏
HIC chromatography column 1 ❏
Column end cap 1 ❏
Waste beaker or tube 1 ❏
Instructors workstation items
Binding buffer 1 bottle ❏
Equilibration buffer 1 bottle ❏
Centrifuge 1 ❏
UV light 1–4 ❏
36

Laboratory Procedure for Lesson 4
1. Remove your microcentrifuge tube from the freezer and thaw it out using hand warmth.
Place the tube in the centrifuge and pellet the insoluble bacterial debris by spinning for 10
minutes at maximum speed. Label a new microcentrifuge tube with your team’s initials.
2. While you are waiting for the centrifuge, prepare the chromatography column. Before per-
forming the chromatography, shake the column vigorously to resuspend the beads. Then
shake the column down one final time, like a thermometer, to bring the beads to the bot-
tom. Tapping the column on the table-top will also help settle the beads at the bottom.
Remove the top cap and snap off the tab bottom of the chromatography column. Allow all
of the liquid buffer to drain from the column (this will take ~3–5 minutes).
3. Prepare the column by adding 2 ml of equilibration buffer to the top of the column, 1 ml at
a time using a well rinsed pipet. Drain the buffer from the column until it reaches the 1 ml
mark which is just above the top of the white column bed. Cap the top and bottom of the
column and store the column at room temperature until the next laboratory period.
4. After the 10 min centrifugation, immediately remove the microcentrifuge tube from the
centrifuge. Examine the tube with the UV light. The bacterial debris should be visible as a
pellet at the bottom of the tube. The liquid that is present above the pellet is called the
supernatant. Note the color of the pellet and the supernatant. Using a new pipet, transfer
250 µl of the supernatant into the new microcentrifuge tube. Again, rinse the pipet well for
the rest of the steps of this lab period.
5. Using the well-rinsed pipet, transfer 250 µl of binding buffer to the microcentrifuge tube
containing the supernatant. Place the tube in the refrigerator until the next laboratory peri-
od.
37
Equilibration buffer (add 2 ml)
1 ml
250 µl
++
Binding buffer (add 250 µl)
+

Lesson 4 Name ________________
Review Questions
1. What color was the pellet in this step of the experiment? What color was the super-
natant? What does this tell you?
2. Why did you discard the pellet in this part of the protein purification procedure?
3. Briefly describe hydrophobic interaction chromatography and identify its purpose in this
lab.
38

Lesson 5
Purification Phase 3
Protein Chromatography
In this final step of purifying the Green Fluorescent Protein, the bacterial lysate you prepared
will be loaded onto a hydrophobic interaction column (HIC). Remember that GFP contains
an abundance of hydrophobic amino acids making this protein much more hydrophobic than
most other bacterial proteins. In the first step, you will pass the supernatant containing the
bacterial proteins and GFP over an HIC column in a highly salty buffer. The salt causes the
three-dimensional structure of proteins to actually change so that the hydrophobic regions of
the protein move to the exterior of the protein and the hydrophilic ("water-loving") regions
move to the interior of the protein.
The chromatography column at your workstation contains a matrix of microscopic hydropho-
bic beads. When your sample is loaded onto this matrix in very salty buffer, the hydrophobic
proteins should stick to the beads. The more hydrophobic the proteins, the tighter they will stick.
The more hydrophilic the proteins, the less they will stick. As the salt concentration is
decreased, the three-dimensional structure of proteins change again so that the hydrophobic
regions of the proteins move back into the interior and the hydrophilic ("water-loving") regions
move to the exterior.
You will use these four solutions to complete the chromatography:
Equilibration buffer—A high salt buffer (2 M (NH
4
)
2
SO
4
)
Binding buffer—A very high salt buffer (4 M (NH
4
)
2
SO
4
)
Wash buffer—A medium salt buffer (1.3 M (NH
4
)
2
SO
4
)
Elution buffer—A very low salt buffer (10 mM Tris/EDTA)
Workstation Check (✔) List
Your Workstation. Make sure the materials listed below are present at your workstation
prior to beginning this lab experiment.
Instructors (Common) Workstation. Materials that should be present at a common location
to be accessed by your group are also listed below.
Your workstation Number (✔)
Collection tubes 3 ❏
Pipets 1 ❏
Microcentrifuge tube rack 1 ❏
Marking pen 1 ❏
Beaker of water for rinsing pipets 1 ❏
HIC chromatography column 1 ❏
Column end cap 1 ❏
Beaker to collect waste 1 ❏
Instructors workstation
Wash buffer 1 vial ❏
Equilibration buffer 1 vial ❏
TE buffer 1 vial ❏
UV light 1–4 ❏
39

Lesson 5 Laboratory Procedure
1. Obtain 3 collection tubes and label them 1, 2, and 3. Place the tubes in a rack. Remove
the cap from the top and bottom of the column and let it drain completely into a liquid
waste container (an extra test tube will work well). When the last of the buffer has
reached the surface of the HIC column bed, gently place the column on collection tube
1. Do not force the column tightly into the collection tubes—the column will not drip.
2. Predict what you think will happen for the following steps and write it along with your
actual observations in the data table on page 42.
3. Using a new pipet, carefully load 250 µl of the supernatant (in binding buffer) into the
top of the column by resting the pipet tip against the side of the column and letting the
supernatant drip down the side of the column wall. Examine the column using the UV
light. Note your observations in the data table. Let the entire volume of supernatant flow
into tube 1.
40
Collection tube 1
2
3
250 µl supernatant in binding buffer
+
Waste tube

4. Transfer the column to collection tube 2. Using the rinsed pipet and the same loading
technique described above, add 250 µl of wash buffer and let the entire volume flow
into the column. As you wait, predict the results you might see with this buffer. Examine
the column using the UV light and list your results on page 42.
5. Transfer the column to tube 3. Using the rinsed pipet, add 750 µl of TE (elution buffer)
and let the entire volume flow into the column. Again, make a prediction and then
examine the column using the UV light. List the results in the data table on page 42.
6. Examine all of the collection tubes using the UV lamp and note any differences in color
between the tubes. Parafilm or plastic wrap the tubes and place in the refrigerator until
the next laboratory period.
41
1
3
Collection tube 2
Wash buffer (250 µl)
Collection tube 3
Add 750 µl TE (elution buffer)
1
2
1
2 3

Lesson 5 Name ________________
Review Questions
1. List your predictions and observations for the sample and what happens to the sample
when the following buffers are added to the HIC column.
Observations
Under UV Light
Collection Tube Number Prediction (column and collection tube)
Tube 1
Sample in binding buffer
Tube 2
Sample with wash buffer
Tube 3
Sample with elution buffer
2. Using the data table above, compare how your predictions matched up with your
observations for each buffer.
a. Binding buffer-
b. Wash buffer-
c. Elution buffer-
3. Based on your results, explain the roles or functions of these buffers. Hint: how does
the name of the buffer relate to its function.
a. Equilibration buffer-
b. Binding buffer-
c. Wash buffer-
d. TE (elution buffer)-
4. Which buffers have the highest salt content and which have the least? How can you
tell?
5. Were you successful in isolating and purifying GFP from the cloned bacterial cells?
Identify the evidence you have to support your answer.
42

Appendix A Glossary of Terms
Agar Provides a solid matrix to support bacterial growth. Contains
nutrient mixture of carbohydrates, amino acids, nucleotides,
salts, and vitamins.
Antibiotic Selection The plasmid used to move the genes into the bacteria also
contain the gene for beta-lactamase which provides resis-
tance to the antibiotic ampicillin. The beta-lactamase protein
is produced and secreted by bacteria which contain the
plasmid. The secreted beta-lactamase inactivates the ampi-
cillin present in the LB/agar, which allows for bacterial
growth. Only bacteria which contain the plasmids, and
express beta-lactamase can survive on the plates which
contain ampicillin. Only a very small percentage of the cells
take up the plasmid DNA and are transformed. Non-trans-
formed cells, cells that do not contain the plasmid, can not
grow on the ampicillin selection plates.
Arabinose A carbohydrate, normally used as source of food by bacte-
ria.
Bacterial Library A collection of
E. coli
that has been transformed with recom-
binant plasmid vectors carrying DNA inserts from a single
species.
Bacterial Lysate Material released from inside a lysed bacterial cell. Includes
proteins, nucleic acids, and all other internal cytoplasmic
constituents.
Beta-Lactamase Beta-lactamase is a protein which provides resistance to
the antibiotic ampicillin. The beta-lactamase protein is pro-
duced and secreted by bacteria which have been trans-
formed with a plasmid containing the gene for
beta-lactamase (
bla
). The secreted beta-lactamase inacti-
vates the ampicillin present in the growth medium, which
allows for bacterial growth and expression of newly acquired
genes also contained on the plasmid
i.e.
GFP.
Biotechnology Applying biology in the real world by the specific manipula-
tion of living organisms, especially at the genetic level, to
produce potentially beneficial products.
Chromatography A process for separating complex liquid mixtures of proteins
or other molecules by passing a liquid mixture over a col-
umn containing a solid matrix. The properties of the matrix
can be tailored to allow for the selective separation of one
kind of molecule from another. Properties include solubility,
molecular size, and charge.
43

Cloning When a population of cells is prepared by growth from a
single cell, all the cells in the population will be genetically
identical. Such a population is called “clonal”. The process
of creating a clonal population is called “cloning”. Identical
copies of a specific DNA sequence, or gene, can be accom-
plished following mitotic division of a transformed host cell.
Colony A clump of genetically identical bacterial cells growing on
an agar plate. Because all the cells in a single colony are
genetically identical, they are called clones.
Centrifugation Spinning a mixture at very high speed to separate heavy
and light particles. In this case, centrifugation results in a
“pellet” found at the bottom of the tube, and a liquid “super-
natant” that resides above the pellet.
Culture Media The liquid and solid media are referred to as LB (named
after Luria-Bertani) broth and agar are made from an extract
of yeast and an enzymatic digest of meat byproducts which
provides a mixture of carbohydrates, amino acids,
nucleotides, salts, and vitamins, all of which are nutrients
for bacterial growth. Agar, which is from seaweed, poly-
merizes when heated to form a solid gel (very analogous
to Jell-O), and functions to provide a solid support on which
to culture the bacteria.
DNA Library When DNA is extracted from a given cell type, it can be cut
into pieces and the pieces can be cloned en masse into a
population of plasmids. This process produces a popula-
tion of hybrid
i.e.
recombinant DNAs. After introducing these
hybrids back into cells, each transformed cell will have
received and propagated one unique hybrid. Every hybrid
will contain the same vector DNA but a different “insert”
DNA.
If there are 1,000 different DNA molecules in the original
mixture, 1,000 different hybrids will be formed; 1,000 dif-
ferent transformant cells will be recovered, each carrying
one of the original 1,000 pieces of genetic information. Such
a collection is called a DNA library. If the original extract
came from human cells, the library is a human library.
Individual DNAs of interest can be fished out of such a
library by screening the library with an appropriate probe.
Genetic Engineering The manipulation of an organism’s genetic material (DNA)
by introducing or eliminating specific genes.
44

Gene Regulation Gene expression in all organisms is carefully regulated to
allow for differing conditions and to prevent wasteful over-
production of unneeded proteins. The genes involved in the
transport and breakdown of food are good examples of
highly regulated genes. For example, the simple sugar, ara-
binose, can be used as a source of energy and carbon by
bacteria. The bacterial enzymes that are needed to break-
down or digest arabinose for food are not expressed in the
absence of arabinose but are expressed when arabinose
is present in the environment. In other words when arabi-
nose is around the genes for these digestive enzymes are
turned on. When arabinose runs out these genes are turned
back off. See Appendix D for a more detailed explanation of
the role that arabinose plays in the regulation and expres-
sion of the green fluorescent protein gene.
Green Fluorescent Protein Green Fluorescent Protein (GFP) was originally isolated from
the bioluminescent jellyfish,
Aequorea victoria
. The gene for
GFP has recently been cloned. The unique three-dimen-
sional conformation of GFP causes it to
resonate when exposed to ultraviolet and give off energy in
the form of visible green light.
Lysozyme Enzyme needed to lyse, or break open bacteria cell walls.
The enzyme occurs naturally in human tears, acting as a
bactericidal agent to help prevent bacterial eye infections.
Lysozyme gets its name from its ability to lyse bacteria.
Pellet In centrifugation, the heavier particles such as bacteria or
the cellular membranes and other debris of lysed bacteria
are found at the bottom of a microfuge tube in a pellet.
Plasmid A circular DNA molecule, capable of autonomous replica-
tion, carrying one or more genes for antibiotic resistance
proteins.
pGLO Plasmid containing the GFP sequence and ampicillin resis-
tance gene which codes for Beta-lactamase.
Recombinant DNA Technology The process of cutting and recombining DNA fragments as
a means to isolate genes or to alter their structure and func-
tion.
Screening Process of identifying wanted bacteria from a bacterial
library.
Sterile Technique Minimizing the possibility of outside bacterial contamination
during an experiment through observance of cleanliness
and using careful laboratory techniques. See Appendix C.
Streaking Process of passing an inoculating loop with bacteria on it
across an agar plate.
Supernatant Liquid containing cellular debris that are lighter than the
debris in the pellet formed after centrifugation.
45

Vector An autonomously replicating DNA molecule into which foreign DNA
fragments are inserted and then propagated in a host cell (
i.e.
plas-
mid).
46

Appendix B
Basic Molecular Biology Concepts and Terminology
A study of the living world reveals that all living organisms organize themselves in some
unique fashion. A detailed blueprint of this organization is passed on to offspring.
Cells are the smallest functional units capable of independent reproduction. Many bacteria, for
instance, can survive as single cells. The chemical molecules within each cell are organized
to perform in concert.
Cells can be grown in culture and harvested
Cells can be gathered from their natural locations and grown inside laboratory containers.
Appropriate food and environment must be provided for the cells to grow. Bacteria and yeast
are very easy to grow in culture. Cells taken from plants, insects, and animals can also be
grown, but are more difficult to care for.
After growth is complete, cells in culture can be harvested and studied.
Cloning
When a population of cells is prepared by growth from a single cell, all the cells in the popu-
lation will be genetically identical. Such a population is called “clonal”. The process of creat-
ing a clonal population is called “cloning”. The purpose of streaking bacteria on agar is to
generate single colonies, each arising from a single cell.
Looking inside cells
The molecules inside a cell each perform a given function. For instance, DNA molecules store
information (like the hard drive in a computer). Proteins are the workhorses of the cell.
To study these molecules we prepare a clonal population from a cell type of interest, break
open the cells, and sort the contents. For instance, it is fairly easy to separate all the proteins
from all the DNA molecules.
Purifying a single species of protein out of the mixture of proteins found inside a cell type is
also possible. Each type of protein has unique physical and chemical properties. These prop-
erties allow the separation of protein species i.e. based on size, charge, or hydrophobicity, for
instance.
Special molecules, specialized functions
We will take a close look at three very special kinds of molecules found inside cells: DNA,
RNA and proteins. Each of these molecules performs a different function. DNA molecules
are like file cabinets in which information is stored. RNA helps to retrieve and execute the
instructions which are stored in DNA. Proteins are designed to perform chemical chores inside
(and often outside) the cell.
DNA: The universal template for biological information
The master “script” for each organism is encoded within its deoxyribonucleic acid (DNA). The
information within the DNA molecule/s of each cell is sufficient to initiate every function that
cell will perform.
DNA molecules are very long chains composed of repeating subunits. Each subunit
(“nucleotide”) contains one of four possible bases protruding from its side:
ADENINE (“A”) CYTOSINE (“C”)
THYMINE (“T”) GUANINE (“G”)
47

Since nucleotides are joined head-to-tail, a long strand of DNA essentially consists of a chem-
ical backbone with bases protruding along its side. The information carried by this molecule
is encoded in the sequence of As, Gs, Cs and Ts along its length.
Some further points to note about DNA structure:
1. Because the subunits of DNA chains are joined head-to-tail, the sequence is directional
i.e. “AACTG” is not the same as “GTCAA”. By convention, we write DNA sequence
from the free 5' end of the backbone and work our way towards the other free end (3').
i.e.
5'...AACTG...3'
2. The protruding bases along the chain are free form spontaneous bonds with available
bases on other DNA strands according to the following rules:
(i) “A” pairs with “T”
(ii) “C” pairs with “G”
Because of these rules, “A” and “T” are said to be complementary bases; “G” and “C”
are also complementary.
(iii) For two DNA strands to pair up, they must be complementary and
run in opposite directions.
i.e. (5'...AGGTC...3’) can pair with (5'...GACCT...3'). These two strands have comple-
mentary sequences. The double-stranded pair is written as follows:
5'...AGGTC...3'
3'...TCCAG...5'
The above molecule contains five base pairs. Indeed, in nature, DNA almost always
occurs in double-stranded form, the two strands containing complementary sequences.
3. DNA molecules are typically thousands, sometimes millions of base pairs long!!
Sometimes the two ends of a DNA molecule are joined to form circular DNA.
4. Double-stranded DNA, in its native form, occurs as a coiled spring, or helix. Because it
is two-stranded, it is often referred to as a double helix.
The architecture of DNA allows for a very simple strategy during reproduction: The two strands
of each DNA molecule unwind and “unzip”; then, each strand allows a new complementary
copy of itself to be made by an enzyme called “DNA polymerase”. This results in two daugh-
ter molecules, each double-stranded, and each identical to the parent molecule.
Proteins and RNA are the workhorses of the cell
The biochemistry of life requires hundreds of very specific and efficient chemical interactions,
all happening simultaneously. The major players in these interactions are short-lived protein
and RNA molecules which can work together or independently to serve a variety of functions.
Like DNA, RNA and proteins are also long chains of repeating units.
RNA
RNA (ribonucleic acid), like DNA, consists of four types of building blocks strung together in
a chain. It differs from DNA in the following respects:
48

The four bases in RNA are “A”, “G”, “C” and “U” (uracil); the pairing rules are the same
as for DNA except that “A” pairs with “U”. Although RNA can pair with complementary
RNA or DNA, in cells RNA is usually single-stranded. The sugar in the RNA backbone
is ribose, not deoxyribose. RNA molecules are generally short, compared to DNA
molecules; this is because each RNA is itself a copy of a short segment from a DNA
molecule. The process of copying segments of DNA into RNA is called transcription,
and is performed by a protein called RNA polymerase.
Proteins
Proteins (more precisely, polypeptides) are also long, chain-like molecules but more structurally
diverse than either DNA or RNA. This is because the subunits of proteins called amino acids,
come in twenty different types. The exact sequence of amino acids along a polypeptide chain
determines how that chain will “fold” into a compact structure. The precise 3-dimensional fea-
tures of this structure, in turn, determine its function.
What a protein will do depends on the exact sequence of its amino acids.
In most cases, a protein will perform a single function. Very diverse functions can be per-
formed by proteins: Some proteins, called “enzymes”, act as catalysts in chemical reactions;
some carry signals from one part of a cell to another - or, in the case of “hormones”, from
one cell to another; some proteins (“antibodies”) have the task of fighting intruders; many
become integral parts of the various physical structures inside cells; and still others (“regula-
tory proteins”) police various activities within cells so as to keep them within legal limits.
Linear code, three-dimensional consequences
DNA is the primary depot for information in living systems. As mentioned, this information is
linear i.e. encoded in the sequence of “A”, “G”, “C”, “T” building blocks along the DNA molecule.
This linear code can be passed on to offspring — because DNA can make exact copies of
itself.
Short segments of each DNA molecule are chosen for transcription at any given time. These
segments are called “genes”. The enzyme RNA polymerase copies the entire segment, base
by base, assembling an RNA molecule which contains a sequence of “A”, “G”, “C” and “U”
exactly complementary to the DNA sequence of the transcribed gene.
In addition to providing a master template for copying RNAs, DNA also contains sequence
information which tells the RNA polymerase where to start transcribing a gene (“promoter”) and
where to stop; how many copies it should make and when; and it can even embed certain infor-
mation within the RNA sequence to determine the longevity and productivity of that RNA!
There are three major classes of RNAs copied off DNA templates: messenger RNAs, or
mRNAs, which relay the sequence information required for assembling proteins; transfer
RNAs, or tRNAs, which work in the assembly line for proteins; and RNAs which perform struc-
tural functions. For example, ribosomal RNAs, or rRNAs, help build the scaffolding for ribo-
somes, the factories where proteins are assembled.
mRNAs carry the sequence information for making proteins. Ribosomes read this sequence
of nucleotides, by a process called “translation” into a sequence of amino acids. How is this
accomplished? There are only four kinds of nucleotides, but twenty kinds of amino acids!
During translation, the ribosome reads 3 nucleotides at a time and assigns an amino acid to
each successive “triplet”. [NOTE: Triplets are often referred to as “codons”.] Each amino acid
is then attached to the end of the growing protein chain. There are 64 possible triplets, or
codons. Thus, the linear information residing in DNA is used to assemble a linear sequence
of amino acids in a protein. This sequence, in turn, will determine the way that protein will
fold into a precise shape with characteristic chemical properties.
49

In summary, the primary transfer of information within cells follows the order:
DNA >>> RNA>>> PROTEIN>>>>TRAIT
Although the information itself is linear, the implications are 3-dimensional. A fundamental
assumption of recombinant DNA technology is that permanent and desirable changes in the
functioning of living cells can be accomplished by changing the linear sequence of their DNA.
Genes are discrete files of DNA information
A gene is a segment within a DNA molecule singled out for copying into RNA. Directly or indi-
rectly, this RNA will perform a function. It is convenient to think of a gene, therefore, as a unit
of function.
Many traits, such as bacterial resistance to an antibiotic, are governed by single genes. Most
traits—such as the color of a rose, or the shape of a nose—are governed by several genes
acting in concert.
Genes can vary in length: Some are only a few hundred base pairs long; some can be tens
of thousands of base pairs long. A DNA molecule may carry from a handful to thousands of
genes. A cell, in turn, may contain one or several DNA molecules (chromosomes). Thus the
number of genes in a cell can vary greatly. E.coli, a bacterium, contains one DNA molecule
with about five thousand genes on it. A human cell contains 46 DNA molecules carrying a
total of about 100,000 genes.
All genes in a given cell are not copied into RNA (i.e. “expressed”) at the same time or at the
same rate. Thus, when speaking of gene function, one refers to its “expression level”. This rate
can be controlled by the cell, according to predetermined rules which are themselves written
into the DNA.
An example: The cells in our bodies (all 100 trillion of them) each contain identical DNA
molecules. Yet liver cells, for example, express only those genes required for liver function,
whereas skin cells express a quite different subset of genes!
DNA can be cut into pieces with restriction enzymes
Restriction enzymes are proteins made by bacteria as a defense against foreign, invading
DNA (for example, viral DNA). Each restriction enzyme recognizes a unique sequence of typ-
ically 4–6 base pairs, and will cut any DNA whenever that sequence occurs.
For example, the restriction enzyme BamHI recognizes the sequence (5'..GGATCC..3') and
cuts the DNA strand between the two G nucleotides in that sequence.
Restriction enzymes will cut DNA from any source, provided the recognition sequence is pre-
sent. It does not matter if the DNA is of bacterial, plant or human origin.
Pieces of DNA can be joined by DNA ligase
DNA ligase is an enzyme that “glues” pieces of DNA together, provided the ends are com-
patible.
Thus, a piece of human or frog or tomato DNA cut with BamHI can be easily joined to a piece
of bacterial DNA also cut with BamHI. This allows the creation of recombinant DNAs i.e.
hybrids, created by joining pieces of DNA from two different sources.
Genes can be cut out of human DNA, or plant DNA, and placed inside bacteria. For example,
the human gene for the hormone insulin can be put into bacteria. Under the right conditions,
these bacteria can make authentic human insulin!
50

Plasmids are small circular pieces of DNA
Plasmids are small circular DNAs found inside some bacterial cells. They replicate their own
DNA by “borrowing” the cells’ polymerases. Thus they can persist indefinitely inside cells with-
out doing very much work of their own!
Because of their small size, plasmid DNAs are easy to extract and purify from bacterial cells.
When cut with a restriction enzyme, they can be joined to foreign DNAs - from any source -
which have been cut with the same enzyme.
The resulting hybrid DNAs can be re-introduced into bacterial cells by a procedure called
transformation. Now the hybrid plasmids can perpetuate themselves in the bacteria just as
before except that the foreign DNA which was joined to it is also being perpetuated. The for-
eign DNA gets a free ride, so to speak.
Every hybrid plasmid now contains a perfect copy of the piece of foreign DNA originally joined
to it. We say that foreign piece of DNA has been “cloned”; the plasmid which carried the for-
eign DNA is called a cloning vehicle or vector.
In addition to their usefulness for cloning foreign genes, plasmids sometimes carry genes of
their own. Bacteria die when exposed to antibiotics. However, antibiotic-resistance genes
allow bacteria to grow in the presence of an antibiotic such as ampicillin. Such genes are
often found on plasmids. When foreign DNA is inserted into such plasmids, and the hybrids
introduced into bacterial cells by transformation, it is easy to select those bacteria that have
received the plasmid—because they have acquired the ability to grow in the presence of the
antibiotic, whereas all other bacterial cells are killed.
DNA libraries
When DNA is extracted from a given cell type, it can be cut into pieces and the pieces can be
cloned en masse into a population of plasmids. This process produces a population of hybrid
(i.e. recombinant) DNAs. After introducing these hybrids back into cells, each transformed
cell will have received and propagated one unique hybrid. Every hybrid will contain the same
vector DNA but a different “insert” DNA.
If there are 1,000 different DNA molecules in the original mixture, 1,000 different hybrids will
be formed; 1,000 different transformant cells will be recovered, each carrying one of the orig-
inal 1,000 pieces of genetic information. Such a collection is called a DNA library. If the orig-
inal extract came from human cells, the library is a human library.
Individual DNAs of interest can be fished out of such a library by screening the library with an
appropriate probe.
51

Appendix C
Sterile Technique
With any type of microbiology technique (i.e. working with and culturing bacteria), it is impor-
tant to not introduce contaminating bacteria into the experiment. Because contaminating bac-
teria are ubiquitous and are found on fingertips, bench tops, etc., it is important to minimize
contact with these contaminating surfaces. When students are working with the inoculation
loops, pipets, and agar plates, you should stress that the round circle at the end of the loop,
the tip of the pipet, and the surface of the agar plate should not be touched or placed onto con-
taminating surfaces. While some contamination will not likely ruin the experiment, students
would benefit from an introduction to the idea of sterile technique. Using sterile technique is
also an issue of human cleanliness and safety.
52

53
Appendix D
Gene Regulation. One Gene: One Protein.
Our bodies contain thousands of different proteins which perform many different jobs. Digestive
enzymes are proteins; some of the hormone signals that run through our bodies and the anti-
bodies protecting us from disease are proteins. The information for assembling a protein is car-
ried in our DNA. The section of DNA which contains the code for making a protein is called a
gene. There are over 100,000 genes in the human genome. Each gene codes for a unique
protein: one gene-one protein. The gene which makes a digestive enzyme in your mouth is
different from one which makes an antibody or the pigments that color your eyes.
Organisms regulate expression of their genes and ultimately the amounts and kinds of pro-
teins present within their cells for a myriad of reasons including developmental, cellular spe-
cialization and adaptation to the environment. Gene regulation not only allows for adaptation
to differing conditions, but also prevents wasteful overproduction of unneeded proteins which
would put the organism at a competitive disadvantage. The genes involved in the transport and
breakdown (catabolism) of food are good examples of highly regulated genes. For example,
the sugar arabinose is both a source of energy and a source of carbon. E. coli bacteria pro-
duce three enzymes (proteins) needed to digest arabinose as a food source. The genes which
code for these enzymes are not expressed when arabinose is absent, but they are expressed
when arabinose is present in their environment. How is this so?
Regulation of the expression of proteins often occurs at the level of transcription from DNA into
RNA. This regulation takes place at a very specific location on the DNA template, called a
promoter, where RNA polymerase sits down on the DNA and begins transcription of the gene.
In bacteria, groups of related genes are often clustered together and transcribed into RNA
from one promoter. These clusters of genes controlled by a single promoter are called oper-
ons.
The three genes (
ara
B
, ara
A
and ara
D) that code for three digestive enzymes involved in the
breakdown of arabinose are clustered together in what is known as the arabinose operon.
These three proteins are dependent on initiation of transcription from a single promoter, (PBAD).
Transcription of these three genes requires the simultaneous presence of the DNA template
(promoter and operon), RNA polymerase, a DNA binding protein called AraC, and arabinose.
AraC binds to the DNA at the binding site for the RNA polymerase (the beginning of the ara-
binose operon). When arabinose is present in the environment, bacteria take it up. Once
inside, the arabinose interacts directly with AraC which is bound to the DNA. The interaction
causes AraC to change its shape which in turn promotes (actually helps) the binding of RNA
polymerase and the three genes B, A, and D, are transcribed. Three enzymes are produced,
they do their job, and eventually the arabinose runs out. In the absence of arabinose, the
AraC returns to its original shape and transcription is shut off.
The DNA code of the pGLO plasmid has been engineered to incorporate aspects of the ara-
binose operon. Both the promoter (PBAD) and the
ara
C gene are present. However, the genes
which code for arabinose catabolism,
ara
B, A and D, have been replaced by the single gene
which codes for the Green Fluorescent Protein (GFP). Therefore, in the presence of arabinose,
AraC protein promotes the binding of RNA polymerase and GFP is produced. Cells fluoresce
a brilliant green color as they produce more and more protein. In the absence of arabinose,
AraC no longer facilitates the binding of RNA polymerase and the GFP gene is not tran-
scribed. When the GFP protein is not made, bacteria colonies will appear to have a wild type
(natural) phenotype - of white colonies with no florescence.
This is an excellent example of the central dogma of molecular biology in action;
DNA>RNA>PROTEIN>TRAIT.

The Arabinose Operon
Expression of Green Fluorescent Protein
54
araB
arabinose
araA araDaraC
araC
araC
araB
RNA Pol
P
BAD
P
BAD
RNA Pol
araA araD
araB araA araD
araC
RNA Pol
araB
m RNA
araD
GFP
arabinose
araC
araC
araC
araC
GFP
RNA Pol
P
BAD
RNA Pol
RNA Pol
GFP
GFP
m RNA

Parafilm is a trademark of American National Can Co.
Jell-O is a trademark of Kraft Foods, Inc.
55

4006099 Rev C
