
Medical Device
Medical Device Coordination Group Document MDCG 2019-11
MDCG 2019-11
Guidance on Qualification and Classification
of Software in Regulation (EU) 2017/745 – MDR
and Regulation (EU) 2017/746 – IVDR
October 2019
This document has been endorsed by the Medical Device Coordination Group
(MDCG) established by Article 103 of Regulation (EU) 2017/745. The MDCG is
composed of representatives of all Member States and it is chaired by a
representative of the European Commission.
The document is not a European Commission document and it cannot be regarded
as reflecting the official position of the European Commission. Any views expressed
in this document are not legally binding and only the Court of Justice of the European
Union can give binding interpretations of Union law.

Page 1 of 28
Guidance on Qualification and
Classification of Software in
Regulation (EU) 2017/745 –
MDR and
Regulation (EU) 2017/746 – IVDR
Guidance on
Qualification and
Classification of
Software
O
ctober 201
9
Page 2 of 28
Table of Contents
1. Scope and purpose of this document 3
2. Definitions and abbreviations 3
3. Qualification 6
3.1. Introduction to qualification criteria 6
3.2. Medical Device Software (MDSW) 7
3.3. ‘Software driving or influencing the use of a medical device’ 8
3.4. Qualification criteria of MDSW as an in vitro diagnostic medical device 10
4. Classification of MDSW per MDR 2017/745 12
4.1. Implementing Rules 12
4.2. Classification Rules 12
5. Classification and implementing rules per IVDR 2017/746 15
5.1. Implementing Rules: 15
5.2. Classification Rules: 15
6. Considerations on placing on the market and conformity assessment of MDSW 16
6.1. Option 1: as a medical device in its own right 16
6.2. Option 2: as an integral component/part of a device 17
7. Modules 17
8. Consideration of changes to an MDSW 18
9. Annex I: Illustrative examples of qualification of software used in the healthcare
environment 18
10. Annex II - Qualification examples of Medical Device Software (MDSW) according to
Figures 1 and 2 24
11. Annex III - Usability of the IMDRF risk classification framework in the context of the
MDR 26
12. Annex IV – Classification examples 27

Page 3 of 28
1. Scope and purpose of this document
This document, which primarily targets medical software manufacturers, defines the criteria for the
qualification of software falling within the scope of the new medical devices regulations
1
and provides
guidance on the application of classification criteria for software under Regulation (EU) 2017/745 –
MDR and Regulation (EU) 2017/746 – IVDR.
2
The guidance also provides information related to
placing on the market. The classification criteria (classification rules) are set out in Annex VIII of the
Medical Devices Regulation (EU) 2017/745 (MDR) and Annex VIII of the In vitro Diagnostic
Medical Devices Regulation (EU) 2017/746 (IVDR).
The criteria specified in this document shall also apply to applications (commonly referred to as apps),
may they be operating on a mobile phone, in the cloud or on other platforms.
2. Definitions and abbreviations
Intended purpose:
According to Regulation (EU) 2017/745 – MDR, “Intended purpose” means the use for which a
device is intended according to the data supplied by the manufacturer on the label, in the instructions
for use or in promotional or sales materials or statements and as specified by the manufacturer in the
clinical evaluation;
3
According to Regulation (EU) 2017/746 – IVDR, “Intended purpose” means the use for which a
device is intended according to the data supplied by the manufacturer on the label, in the instructions
for use or in promotional or sales materials or statements or as specified by the manufacturer in the
performance evaluation;
4
Accessory:
According to Regulation (EU) 2017/745 – MDR, “Accessory for a medical device” means an article
which, whilst not being itself a medical device, is intended by its manufacturer to be used together
with one or several particular medical device(s) to specifically enable the medical device(s) to be used
in accordance with its/their intended purpose(s) or to specifically and directly assist the medical
functionality of the medical device(s) in terms of its/their intended purpose(s);
5
According to Regulation (EU) 2017/746 – IVDR, “Accessory for an in vitro diagnostic medical
device” means an article which, whilst not being itself an in vitro diagnostic medical device, is
intended by its manufacturer to be used together with one or several particular in vitro diagnostic
medical device(s) to specifically enable the in vitro diagnostic medical device(s) to be used in
accordance with its/their intended purpose(s) or to specifically and directly assist the medical
functionality of the in vitro diagnostic medical device(s) in terms of its/their intended purpose(s);
6
Note: Software accessory may be driving or influencing the use of a medical device.
Note: Manufacturers must describe in the technical documentation accessories of a medical device or
an in vitro diagnostic medical device which are intended to be used in combination with it; and in case
1
The use of “The Medical Devices Regulations” from here on out refers to both Regulation (EU) 2017/745 – MDR and
Regulation (EU) 2017/746 – IVDR.
2
It shall be noted that the term “standalone software” which was used in the text of the medical device directives, is no
longer used in the context of the Medical Device Regulation. The rationale of the change is that software should be qualified
and classified depending on its intended purpose uniquely, regardless of its location.
3
Article 2(12) of Regulation (EU) 2017/745 – MDR
4
Article 2(12) of Regulation (EU) 2017/746 – IVDR
5
Article 2(2) of Regulation (EU) 2017/745 – MDR
6
Article 2(4) of Regulation (EU) 2017/746 – IVDR

Page 4 of 28
of medical devices they must include in the instructions for use information allowing the selection of
the corresponding software and accessories.
7
Placing on the market:
According to Regulation (EU) 2017/745 – MDR, “Placing on the market” means the first making
available of a device, other than an investigational device, on the Union market;
8
According to Regulation (EU) 2017/746 – IVDR, “Placing on the market” means the first making
available of a device, other than a device for performance study, on the Union market;
9
Putting into service:
According to Regulation (EU) 2017/745 – MDR, “Putting into service” means the stage at which a
device, other than an investigational device, has been made available to the final user as being ready
for use on the Union market for the first time for its intended purpose;
10
According to Regulation (EU) 2017/746 – IVDR, “Putting into service” means the stage at which a
device, other than a device for performance study, has been made available to the final user as being
ready for use on the Union market for the first time for its intended purpose;
11
Medical device:
“medical device” means any instrument, apparatus, appliance, software, implant, reagent, material or
other article intended by the manufacturer to be used, alone or in combination, for human beings for
one or more of the following specific medical purposes:
- diagnosis, prevention, monitoring, prediction, prognosis, treatment or alleviation of disease,
- diagnosis, monitoring, treatment, alleviation of, or compensation for, an injury or disability,
- investigation, replacement or modification of the anatomy or of a physiological or
pathological process or state,
- providing information by means of in vitro examination of specimens derived from the human
body, including organ, blood and tissue donations,
and which does not achieve its principal intended action by pharmacological, immunological or
metabolic means, in or on the human body, but which may be assisted in its function by such means.
The following products shall also be deemed to be medical devices:
- devices for the control or support of conception;
- products specifically intended for the cleaning, disinfection or sterilisation of devices as
referred to in Article 1(4) and of those referred to in the first paragraph of this point.
12
Active medical device:
“active device” means any device, the operation of which depends on a source of energy other than
that generated by the human body for that purpose, or by gravity, and which acts by changing the
density of or converting that energy. Devices intended to transmit energy, substances or other elements
between an active device and the patient, without any significant change, shall not be deemed to be
active devices. Software shall also be deemed to be an active device;
13
7
Article 32.2(c), Annex I, Chapter III Section 23.4(f), Annex II Section 1.1(h) of Regulation (EU) 2017/745 – MDRArticle
29.2(c) and Annex II Section 1.1(m) of Regulation (EU) 2017/746 – IVDR
8
Article 2(28) of Regulation (EU) 2017/745 – MDR
9
Article 2(21) of Regulation (EU) 2017/746 – IVDR
10
Article 2(29) of Regulation (EU) 2017/745 – MDR
11
Article 2(22) of Regulation (EU) 2017/746 – IVDR
12
Article 2(1) of Regulation (EU) 2017/745 – MDR
13
Article 2(4) of Regulation (EU) 2017/745 – MDR

Page 5 of 28
In vitro diagnostic medical device:
“In vitro diagnostic medical device” means any medical device which is a reagent, reagent product,
calibrator, control material, kit, instrument, apparatus, piece of equipment, software or system,
whether used alone or in combination, intended by the manufacturer to be used in vitro for the
examination of specimens, including blood and tissue donations, derived from the human body, solely
or principally for the purpose of providing information on one or more of the following:
- concerning a physiological or pathological process or state;
- concerning congenital physical or mental impairments;
- concerning the predisposition to a medical condition or a disease;
- to determine the safety and compatibility with potential recipients;
- to predict treatment response or reactions;
- to define or monitoring therapeutic measures.
Specimen receptacles shall also be deemed to be in vitro diagnostic medical devices;
14
Software:
For the purpose of this guidance, “software” is defined as a set of instructions that processes input data
and creates output data.
Input data:
Any data provided to software in order to obtain output data after computation of this data can be
considered as input data. Input data examples (non-exhaustive):
- Data given through the use of a human data-input device such as a keyboard, mouse, stylus, or
touch screen;
- Data given through speech recognition;
- Digital document: formatted for general purpose such as Word file or pdf file or jpeg image,
formatted for medical purpose such as DICOM file or ECG records or Electronic Health
Record, unformatted document. Note that digital documents have to be differentiated from
software able to read such documents;
- Data received from/transmitted by devices.
Output data:
Any data produced by a software can be considered as an output data. Output data examples (non-
exhaustive):
- Screen display data (such as layout with number, characters, picture, graphics, etc.);
- Print data (such as layout with number, characters, picture, graphics, etc.);
- Audio data;
- Digital document (formatted for a general purpose such as Word file or pdf file or jpeg image,
or formatted for medical purpose such as DICOM file or ECG records or Electronic Health
Record, unformatted document).
- Haptic buzzing as an alternative to audio sound
Software driving or influencing the use of a device
Software which is intended to drive or influence the use of a (hardware) medical device and does not
have or perform a medical purpose on its own, nor does it create information on its own for one or
more of the medical purposes described in the definition of a medical device or an in vitro diagnostic
medical device. This software can, but is not limited to:
(a) operate, modify the state of, or control the device either through an interface (e.g., software,
hardware) or via the operator of this device
(b) or supply output related to the (hardware) functioning of that device
Note: Software driving or influencing the use of a (hardware) medical device may be qualified as an
accessory for a (hardware) medical device.
14
Article 2(2) of Regulation (EU) 2017/746 – IVDR

Page 6 of 28
Medical Device Software (MDSW)
Medical device software is software that is intended to be used, alone or in combination, for a purpose
as specified in the definition of a “medical device” in the medical devices regulation
15
or in vitro
diagnostic medical devices regulation.
16
3. Qualification
3.1. Introduction to qualification criteria
The purpose of this section is to clarify what software is in itself subject to the medical devices
regulations.
Software must have a medical purpose on its own to be qualified as a medical device software
(MDSW). It should be noted that the intended purpose as described by the manufacturer of the
software is relevant for the qualification and classification of any device.
In order to be qualified as medical device software, the product must first fulfil the definition of
software according to this guidance and the definition of a medical device according to Article 2(1) of
Regulation (EU) 2017/745 – MDR. To be qualified as an in vitro diagnostic medical device software,
the product must additionally fulfil the definition of an in vitro diagnostic medical device according to
Article 2(2) of Regulation (EU) 2017/746 – IVDR.
Where a given product does not fall under the definition of a medical device, or is excluded by the
scope of the Medical Devices Regulations, other Community and/or national legislation may be
applicable. Software that does not meet the definition of a medical device or an in vitro diagnostic
medical device (i.e. software that is not MDSW) but is intended by the manufacturer to be an
accessory for a medical device, or an in vitro diagnostic medical device, falls respectively under the
scope of the Regulation (EU) 2017/745 – MDR or Regulation (EU) 2017/746 – IVDR.
Software can directly control a (hardware) medical device (e.g. radiotherapy treatment software), can
provide immediate decision-triggering information (e.g. blood glucose meter software), or can provide
support for healthcare professionals (e.g. ECG interpretation software).
It is important to clarify that not all software used within healthcare is qualified as a medical device.
For example, “Simple search”, which refers to the retrieval of records by matching record metadata
against record search criteria or to the retrieval of information does not qualify as medical device
software (e.g. library functions).
However, software which is intended to process, analyse, create or modify medical information may
be qualified as a medical device software if the creation or modification of that information is
governed by a medical intended purpose. For example, the software which alters the representation of
data for a medical purpose would qualify as a medical device software. (e.g. “searching image for
findings that support a clinical hypothesis as to the diagnosis or evolution of therapy” or “software
which locally amplifies the contrast of the finding on an image display so that it serves as a decision
support or suggests an action to be taken by the user”). However, altering the representation of data for
embellishment/cosmetic or compatibility purposes does not readily qualify the software as medical
device software.
Software intended for non-medical purposes (excluding MDR Annex XVI devices), such as invoicing
or staff planning, does not qualify as a medical device software. These software do not fall under the
Medical Devices Regulations.
15
Article 2(1) of Regulation (EU) 2017/745 – MDR
16
Article 2(2) of Regulation (EU) 2017/746 – IVDR

Page 7 of 28
A task such as e-mailing, web or voice messaging, data parsing, word processing, and back-up is by
itself not considered as having a medical purpose.
Additionally, software may run on different operating systems or in virtual environments. These
operating systems or virtual environments do not impact the qualification criteria.
It must be highlighted that the risk of harm to patients, users of the software, or any other person,
related to the use of the software within healthcare, including a possible malfunction is not a criterion
on whether the software qualifies as a medical device.
3.2. Medical Device Software (MDSW)
Medical device software is software that is intended to be used, alone or in combination, for a
purpose as specified in the definition of a “medical device” in the MDR or IVDR, regardless of
whether the software is independent or driving or influencing the use of a device.
Note 1: MDSW may be independent, by having its own intended medical purpose and thus meeting
the definition of a medical device or in vitro diagnostic medical device on its own (i.e. alone)
MDSW that uses maternal parameters such as age, concentration of serum markers and information
obtained through foetal ultrasound examination for evaluating the risk of trisomy 21.
MDSW that receives measurements from transrectal ultrasound findings, age, and in vitro diagnostic
instruments and calculates a patient’s risk of developing prostate cancer.
Mass Spectrometry MDSW intended to analyse LC-MS/MS data to be used for microorganism
identification and detection of antibiotic resistance.
MDSW smartwatch app, which is intended to send alarm notifications to the user and/or health
practitioner when it recognises irregular heartbeats for the purpose of detecting cardiac arrhythmia.
Note 2: If the software drives or influences a (hardware) medical device and also has a medical
purpose, then it is qualified as a MDSW
Melanoma image analysis software intended to drive a near-infrared laser light scanner.
MDSW intended to measure and transmit blood glucose levels, calculate insulin dose required and
drive the insulin pump to administer the calculated dosage (closed loop insulin delivery system).
Note 3: Software may be qualified as MDSW regardless of its location (e.g. operating in the cloud, on
a computer, on a mobile phone, or as an additional functionality on a hardware medical device).
MDSW that is intended to operate a point of care test from a remote location.
Note 4: MDSW may be intended to be used by healthcare professionals or laypersons (e.g. patients or
other users).
17, 18
MDSW that provides insulin dose recommendations to a patient regardless of the method of delivery
of the prescribed dose, whether via an insulin pump, insulin pen or insulin syringe.
17
Where MDSW is intended to be used by a lay person, the manufacturer shall apply safety and performance requirements
outlined in MDR Annex I. 22 and 23.4 (w); or IVDR Annex I. 9.4 and 20.4.2.
18
MDSW which can be considered as an IVD for self-testing shall be considered as a device intended to be used by
laypersons.

Page 8 of 28
Manufacturers must ensure that all regulatory requirements for placing on the market and conformity
assessment have been fulfilled. As set out in Article 7 of MDR and IVDR, this also entails that any
claims, relating to the intended medical purpose of their MDSW are supported by clinical evidence. If
this is not the case, the software would not meet the requirements of the regulations and therefore may
not be CE marked as a medical device, nor present said claims.
3.3. ‘Software driving or influencing the use of a medical device’
Is software intended to drive or influence the use of a (hardware) medical device and does not have or
perform a medical purpose on its own, nor does it create information on its own for one or more of the
medical purposes described in the definition of a medical device or an in vitro diagnostic medical
device. This software can, but is not limited to:
a) operate, modify the state of, or control the device either through an interface (e.g., software,
hardware) or via the operator of this device
b) or supply output related to the (hardware) functioning of that device
Note: Software that is driving or influencing the use of a medical device is covered by the medical
devices regulations either as a part/component of a device or as an accessory for a medical device.
(refer to Figure 2, box 2).
Software that is intended to be used to operate a clinical chemistry analyser.
Software with built-in electronic controls for IVD quality control procedures. These quality control
procedures are intended to provide users with assurance that the device is performing within
specifications.
Decision steps for qualification of software as MDSW (Figure 1)
Decision step 1: if the product is software according to Section 2 (Definitions and Abbreviations) of
this guidance, then it may be a medical device software, proceed to decision step 2; if the product is
not software according to the definition of this guidance, then it is not covered by this guidance but
may still be covered by the Medical Devices Regulations.
Decision step 2: if the product is an MDR Annex XVI device, or is an accessory for a medical
device
19
, or is software driving or influencing the use of a medical device, then it must be considered
as part of that device in its regulatory process or independently if it is an accessory. If it is not, proceed
to decision step 3.
Decision step 3: if the software does perform an action on data, or performs an action beyond storage,
archival, communication
20
, simple search, lossless compression (i.e. using a compression procedure
that allows the exact reconstruction of the original data) then it may be a medical device software
(Refer to section 3.1 for more guidance on these software functions) proceed to step 4.
Decision step 4: is the action for the benefit of individual patients?
Examples of software which are not considered as being for the benefit of individual patients are those
which are intended only to aggregate population data, provide generic diagnostic or treatment
pathways (not directed to individual patients), scientific literature, medical atlases, models and
templates as well as software intended only for epidemiological studies or registers.
Decision step 5: Is the software medical device software (MDSW) according to the definition of this
guidance?
19
According to Article 2(2) of the Medical Devices Regulation or In Vitro Diagnostic Medical Devices Regulation.
20
Communication: The flow of information from one point, known as the source, to another, the receiver; Source: IEEE
610.10-1994.

Page 9 of 28
Page 10 of 28
3.4. Qualification criteria of MDSW as an in vitro diagnostic medical
device
Medical Device Software (MDSW) fulfilling the definition of an in vitro diagnostic medical device
falls under the in vitro diagnostic medical devices Regulation (EU) 2017/746. Provided that MDSW is
intended specifically by its manufacturer to be used together with an in vitro diagnostic medical device
to enable it to be used in accordance with its intended purpose, this MDSW falls under the scope of the
in vitro diagnostic medical devices regulation and shall be treated as an In Vitro Diagnostic MDSW
(IVD MDSW) in its own right.
In cases when software is driving or influencing the use of a (hardware) device, the software should be
considered as falling under the respective regulation of the driven or influenced (hardware) device.
Software that analyses and interprets the optical density delivered by an ELISA reader, line or spot
pattern of a blot.
Such software takes raw data outputs and use clinical algorithms for diagnostic
and/or prognostic purposes, in which case it qualifies as IVD MDSW.
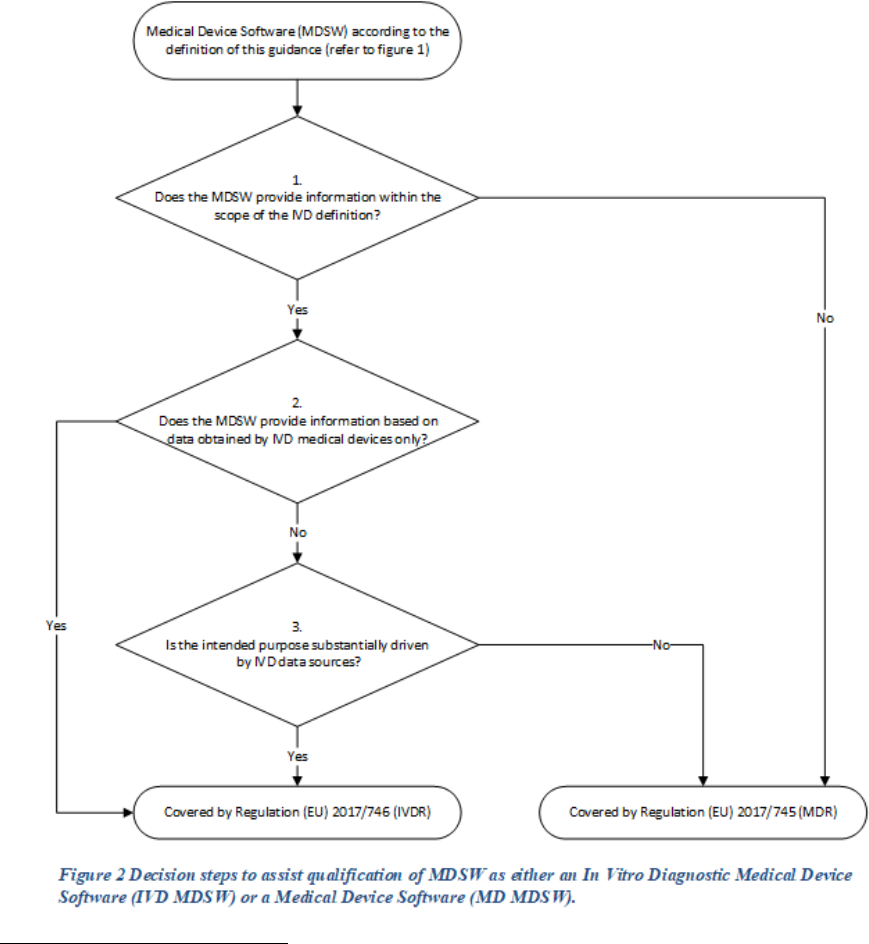
Decision steps for qualification of MDSW as either a medical device or an in vitro
diagnostic medical device (Figure 2)
Decision Step 1: Does the Medical Device Software (MDSW) provide information within the scope
of the in vitro diagnostic medical device definition?
MDSW which provides information according to Regulation (EU) 2017/746 – IVDR Article 2(2) (a)
to (f) should qualify as In Vitro Diagnostic Medical Device Software (IVD MDSW)
(a) concerning a physiological or pathological process or state (by investigation of this process or
state); or
(b) concerning congenital physical or mental impairments
(c) concerning the predisposition to a medical condition or a disease;
(d) to determine the safety and compatibility with potential recipients;
(e) to predict treatment response or reactions;
(f) to define or monitoring therapeutic measures.
A MDSW which falls under the definition set out in EU Article 2 (1) of Regulation (EU) 2017/745 –
MDR should qualify as Medical Device Software (MD MDSW). In specific, the following
considerations should apply on the provision of information by software on:
(g) diagnosis, prevention, monitoring, prediction, prognosis, treatment or alleviation of disease
(h) diagnosis, monitoring, treatment, alleviation of, or compensation for, an injury or disability,
(i) investigation, replacement or modification of the anatomy or of a physiological or
pathological process or state,
(j) control or support of conception;
(k) products specifically intended for the cleaning, disinfection or sterilization of devices as
referred to in Article 1(4) and Annex XVI products.
Decision Step 2: Does the MDSW create information based on data obtained by in vitro diagnostic
medical devices only?
If the information provided is based on data obtained solely from in vitro diagnostic medical devices,
then the software is an in vitro diagnostic medical device and is therefore an IVD MDSW.
If the data analysed is obtained from a combination of both in vitro diagnostic medical devices and
medical devices, proceed to step 3.
Page 11 of 28
Decision Step 3: Is the intended purpose substantially driven by data sources coming from in vitro
diagnostic medical devices? If yes, then the applicable legislation is Regulation (EU) 2017/746. If the
intended purpose is substantially driven by data sources coming from medical devices, then the
applicable legislation is Regulation (EU) 2017/745.
In the condition where the intended purpose of the MDSW output data fulfils both the medical device
and in vitro diagnostic medical device definitions set out in the MDR and IVDR (refer to Decision
Step 2), a weighting of the data sources based on the significance of the information
21
in relation to
fulfilling the intended purpose should be conducted to aid the manufacturer in determining which
regulation to apply.
Annex II of this guidance offers some prescriptive examples of how such a weighting may be
performed.
21
A qualitative approach is intended to drive the weighting of data. The weighing aims to assess the contribution of each data
towards achieving the result.

Page 12 of 28
4. Classification of MDSW per MDR 2017/745
4.1. Implementing Rules
All implementing rules in Annex VIII of Regulation (EU) 2017/745 shall be considered.
Special considerations on Implementing Rule 3.3 and 3.5:
The first sentence of implementing rule 3.3 of Annex VIII clarifies the regime applicable to software
driving or influencing the use of a device:
‘Software, which drives or influences the use of a device, shall fall within the same class as the device’
The second sentence of implementing rule 3.3 of Annex VIII clarifies that the regime applicable to
Independent MDSW:
‘If software is independent of any other device, it shall be classified in its own right’
This rule should also be considered at least as an orientation for finding the correct (minimum)
classification of software which is placed on the market in combination with a (hardware) medical
device. Therefore, Medical Device Software that both achieves its own intended purpose and also
drives or influences the use of a (hardware) device for a medical purpose is classified on its own,
based on the intended purpose achieved. In such a case, however, the risk class shall not be lower than
the risk class of the hardware medical device.
Implementing rule 3.5 of Annex VIII is relevant for all devices and states that
‘If several rules, or if, within the same rule, several sub-rules, apply to the same device based on the
device’s intended purpose, the strictest rule and sub-rule resulting in higher classification will apply’
Melanoma image analysis software intended to be used with a near-infrared laser light scanner,
which is considered class IIa per Rule 10. The software “drives or influences the use of” the near-
infrared laser light scanner as it is intended to take control of the scanner by letting it execute
proprietary multi-exposure programs for the detection of melanoma. As such, implementing rule 3.3
applies. However, Rule 11 would also apply based on the intended medical purpose of the software
e.g. cancer diagnosis. The MDSW would be classified as class III based on Rule 11 (see section
Classification Rules) and per implementing rule 3.5 of Annex VIII.
4.2. Classification Rules
Rules 9, 10 and 12 mainly categorise the risks related to the exchange of energy/substances between
the body and diagnostic or therapeutic active devices, taking into account the different healthcare
situations (condition of patients).
MDSW, in the majority of cases, does not (directly) relate to such risks. It relates to the consequences
of indirect harm from failure to provide correct information.
Therefore, in line with Recital 5 of the Medical Device Regulation and international guidance from the
IMDRF (International Medical Device Regulators Forum)
22
, Rule 11 was introduced into the MDR
and is intended to address the risks related to the information provided by an active device, such as
MDSW. Rule 11, in particular, describes and categorises the significance of the information provided
by the active device to the healthcare decision (patient management) in combination with the
healthcare situation (patient condition).
As software is defined as an active device, for the classification of active (hardware) devices, which
also includes MDSW providing information for patient management, Rules 9, 10, 11, 12, 13, 15 and
22
IMDRF is a voluntary group of medical device regulators from around the world who have come together to build on the
strong foundational work of the Global Harmonization Task Force on Medical Devices (GHTF) and aims to accelerate
international medical device regulatory harmonization and convergence.

Page 13 of 28
22 of Annex VIII MDR 2017/745 should be considered. In line with implementation rule 3.5, the
strictest rule or sub-rule should hence apply.
MDSW should be classified in the same way, regardless of the software's location or the type of
interconnection between the software and a (hardware) device.
4.2.1. Rule 11 – Software for decisions with diagnosis or therapeutic purposes or
software intended to monitor physiological processes
Rule 11 states:
Software intended to provide information which is used to take decisions with diagnosis or
therapeutic purposes is classified as class IIa, except if such decisions have an impact that
may cause:
death or an irreversible deterioration of a person's state of health, in which case it is in class
III; or
a serious deterioration of a person's state of health or a surgical intervention, in which case it
is classified as class IIb.
Software intended to monitor physiological processes is classified as class IIa, except if it is
intended for monitoring of vital physiological parameters, where the nature of variations of
those parameters is such that it could result in immediate danger to the patient, in which case
it is classified as class IIb.
All other software is classified as class I.
The text of Rule 11 can be divided into what are essentially three sub-rules that are applied depending
on the intended use/purpose of the MDSW:
11a: (3 first paragraphs of Rule 11) intended to provide information which is used to take
decisions with diagnostic or therapeutic purposes;
11b: (Paragraph 4 of Rule 11) intended to monitor physiological processes or parameters;
11c: (Paragraph 5 of Rule 11) all other uses.
Sub-rule 11a):
The wording “intended to provide information which is used to take decisions with diagnosis or
therapeutic purposes” describes, in very general terms, the “mode of action” which is characteristic of
all MDSW. Therefore, this sub-rule is generally applicable to all MDSW (excluding those MDSW
that have no medical purpose).
Sub-rule 11a), states that MDSW (which is intended to provide information which is used to take
decisions with diagnosis or therapeutic purposes) is classified as class IIa.
There are two exceptions from sub-rule 11a) that are mainly intended to apply a risk classification
based on the significance of the provided information and the potential impact of an (incorrect)
decision made using information from the MDSW.
23
Accordingly, MDSW that is intended to provide
information which is used to take decisions with diagnosis and therapeutic purposes, is at a higher risk
class where such decisions, if based on incorrect information from the MDSW, are reasonably likely
to have an impact that may cause:
i. death or an irreversible deterioration of a person's state of health, in which case it is in class III;
ii. serious deterioration of a person's state of health or surgical intervention, in which case it is
classified as class IIb.
23
Compare IMDRF/SaMD WG/N12FINAL:2014 which states that there are two major factors that provide adequate
description of the intended use of software: A. - Significance of the information provided by the software to the healthcare
decision and B. - State of the healthcare situation or patient condition.

Page 14 of 28
The MDR contains several references to “serious deterioration of a person’s state of health” and
“surgical intervention”, notably in the vigilance or clinical investigation context. Further horizontal
guidance could be provided in the future and will be available at:
https://ec.europa.eu/growth/sectors/medical-devices/new-regulations/guidance_en
Rule 11 was also introduced to mirror the regulatory guidance developed at international level and
notably in the context of the International Medical Device Regulators Forum (IMDRF). The IMDRF
framework for risk categorisation of software as a medical device (SaMD) (“IMDRF Risk
Framework”) categorises the risk of software based on the combination of the significance of the
information provided by the software to the healthcare decision and the healthcare situation or patient
condition. IMDRF also developed a table for assisting operators in identifying the appropriate risk
category for their own product.
Such a table could provide operators placing MDSW on the EU market with some useful indications
on the risk class applicable to their products as a result of the application of Rule 11 of the MDR. For
this purpose, a table is provided in Annex III to this document which lists the IMDRF risk categories
and the possible corresponding MDR risk classes according to Rule 11 of the MDR. It must be noted
that this table does not take into account MDSW which is Class I.
Sub-rule 11b):
MDSW that is intended to monitor physiological processes will, under most circumstances, provide
“information which is used to take decisions with diagnosis or therapeutic purposes and hence fall
under sub-rule 11a. Sub-rule 11b) should therefore be considered as a specific rule for MDSW
intended only for monitoring purposes. Sub-rule 11b) was introduced to ensure that MDSW which has
the same intended purpose as (hardware) devices which would fall under rule 10, third indent, are in
the same risk class.
However, this sub rule applies to MDSW intended to be used for monitoring any/all physiological
processes and not just vital physiological processes (equivalent to rule 10, third indent).
Vital physiological processes and parameters include, for example, respiration, heart rate, cerebral
functions, blood gases, blood pressure and body temperature.
Sub-rule 11c):
Sub-rule 11c) implies that all other MDSW is classified as class I.
4.2.2. Rule 12 – Active devices intended to administer and/or remove
substances
As software devices cannot physically administer and/or remove substances, please refer to the
implementation rule 3.3 of Annex VIII for MDSW covered by this rule.
4.2.3. Rule 13 – All other active devices
Taking into consideration all implementing and classification rules applicable to active devices, if no
other rule applies, all other active devices are class I.
24
4.2.4. Rule 15 - Devices used for contraception
Rule 15 applies to devices used for contraception or prevention of the transmission of sexually
transmitted diseases. Software used for contraception will be classified as class IIb.
24
Specific implementation or classification rules for active Annex XVI devices (which might also include software) are
expected to be provided together with the relevant Common Specifications for those devices.

Page 15 of 28
4.2.5. Rule 22 – Closed loop systems
Active therapeutic devices with an integrated or incorporated diagnostic function which significantly
determines the patient management by the device, such as closed loop systems or automated external
defibrillators, are classified as class III.
See also implementing rule 3.3 for MDSW covered by this rule.
Further horizontal Guidance on the application of MD classification and implementing rules can be
found at https://ec.europa.eu/growth/sectors/medical-devices/new-regulations/guidance_en. This is
expected to provide useful orientation in relation to the application of non-software specific
classification rules.
5. Classification and implementing rules per IVDR 2017/746
5.1. Implementing Rules:
All implementing rules in Annex VIII of Regulation (EU) 2017/746 shall be considered.
Special considerations on Implementing Rule 1.4 and 1.9:
Implementing rule 1.4 is only applicable for software which drives or influences the use of an in vitro
diagnostic medical device. This rule should also be considered at least as an orientation for finding the
right (minimum) classification of software which is placed on the market in combination with a
(hardware) medical device.
According to the second sentence of implementing rule 1.4, if the software is independent of any other
device, it shall be classified in its own right.
Examples for applying this implementing rule under the in vitro diagnostic medical devices regulation:
• Software that is exclusively intended to drive or influence the use of an instrument intended by
the manufacturer specifically to be used for in vitro diagnostic procedures is classified in the
same class as the instrument.
• A software that is intended to operate (driving) a C-reactive protein (CRP) measuring
analyser from a remote location is classified in the same class as the analyser i.e. if the
analyser is a classified as class A then the software operating the analyser falls into Class A.
• MDSW that integrates genotype of multiple genes to predict risk a disease or medical
condition developing or recurring; this is an independent IVD MDSW and is classified on its
own.
Implementing rule 1.9 states that if several classification rules apply to the same device based on the
devices’ intended purpose, the rule resulting in higher classification will apply. To classify In Vitro
Diagnostic Medical Device Software (IVD MDSW) which is independent of any other device, see the
MDCG Guidance on Classification of IVDs when available at
https://ec.europa.eu/growth/sectors/medical-devices/new-regulations/guidance_en.
5.2. Classification Rules:
In determining the proper classification of MDSW under the IVDR, the manufacturer shall consider
all classification and implementing rules of Annex VIII of the IVD Regulation (EU) 2017/746.
As spelled out by Implementing Rule 1.1 of Annex VIII of Regulation (EU) 2017/746, the application
of the classification rules shall be governed by the intended purpose of the MDSW.

Page 16 of 28
Guidance on the application of the IVD classification and implementing rules can be found at
https://ec.europa.eu/growth/sectors/medical-devices/new-regulations/guidance_en
25
Examples for the classification of MDSW under the IVDR:
• Software intended to be installed on a fully automated enzyme-linked immunosorbent assay
(ELISA) analyser, and intended to determine the Human HbA1c concentration in serum from
the results obtained with a Human HbA1c ELISA, intended to screen for and diagnose
diabetes and monitor diabetic patients, should be in class C per Rule 3(k).
• Software within a PAP stain automated cervical cytology screening system, intended to
classify the PAP cervical smear as either normal or suspicious, should be in class C per Rule
3(h).
• Software for the interpretation of automated readings of line immunoassay for the
confirmation and determination of antibodies to HIV-1, HIV-1 group O and HIV-2 in human
serum and plasma, should be in class D per Rule 1.
• Software that uses maternal parameters such as age, concentration of serum markers and
information obtained through foetal ultrasound examination for evaluating the risk of trisomy
21, should be in class C per Rule 3(l).
Classification examples in Annex IV are provided for guidance purposes and aim to illustrate how
a particular rule may be applied to a device. The indicated classification in the example is not a
confirmation of the final classification of the device, as other rules must also be considered.
6. Considerations on placing on the market and conformity
assessment of MDSW
The type of interconnection between the MDSW and the device (e.g. embedded systems, wires, Wi-Fi,
Bluetooth) does not affect the qualification of the software as a device under the MDR and IVDR (e.g.
whether the software is incorporated in a device or is at a different location). However, MDSW can be
placed on the market in two different ways: as a medical device or in-vitro diagnostic medical device
in its own right or as an integral component or part of a hardware device.
6.1. Option 1: as a medical device in its own right
MDSW may be placed on the market or put into service in its own right.
• MDSW intended to be installed on a fully automated enzyme-linked immunosorbent assay
(ELISA) analyser, and intended to determine the Human HbA1c concentration in serum from
the results obtained with a Human HbA1c ELISA.
• MDSW app that provides a 10-year risk of cardiovascular disease from data input by a lay
user.
• MDSW app that calculates anticoagulant dosage for patients in oral anticoagulant therapy,
from INR test results input by IVD instruments and other manually entered patient data.
• MDSW app that analyses digital images of stained HEp-2 cell substrates from a microscope,
for detecting antinuclear antibody (ANA) patterns to guide confirmatory testing useful in
elucidating a specific clinical diagnosis or prognosis.
Conformity assessment:
25
Please note that at the time of the adoption of this document, the referenced Guidance on the application of IVDR
classification and implementing rules was under finalisation.

Page 17 of 28
MDSW placed on the market as a device or put into service in its own right shall undergo an
appropriate regulatory process that shall take into consideration the qualification, classification and
intended purpose of the MDSW.
6.2. Option 2: as an integral component/part of a device
MDSW may be placed on the market or put into service as an integral component/part of a device.
• MDSW contained within a blood gas analyser that enables a user to run tests on the
instrument.
• MDSW that is part of a handheld hardware device intended for near-patient testing (POCT:
point of care testing) for the determination of the blood glucose concentration.
• A fully automated enzyme-linked immunosorbent assay (ELISA) analyser, composed of
hardware and MDSW, intended to determine the Human HbA1c concentration in serum from
the results obtained with a Human HbA1c ELISA.
• MDSW that is part of a pulse oximeter intended to digitally filter the noise from low perfusion
performance and motion artefacts, to calculate the ratio of red light/infrared light and to use a
lookup table based on Beer-Lambert law to convert the ratio into the oxygen saturation in a
person’s blood (SpO2).
Conformity assessment:
MDSW that is placed on the market or put into service solely as an integral component/part of a
(hardware) device may not have to undergo its own regulatory process.
26
In this case, the MDSW shall
be assessed through the regulatory process applied to the device as a whole, as it is placed on the
market.
Applying the classification rules to these hardware devices, which is de-facto a combination of the
hardware device and the MDSW, requires careful consideration of the intended purpose of the
MDSW. This must also be analysed when later changes to the MDSW are done.
Note: MDSW could be independent under both scenarios described in 6.1 and 6.2, despite the
presentation in which it is placed on the market.
7. Modules
Some medical device software may be segregated into a number of applications where each of these
applications is correlated with a module. Some of these modules have a medical purpose, some not.
Such modules may be intended to cover many needs, e.g.:
• Collect and maintain administrative patient data;
• Keep on file the medical history of the patient;
• Invoicing and other accounting functions;
• Provide a link to the social security system for reimbursement;
• Provide a link to drug prescription systems (with possible link to drug dispensing outlets);
• Provide expert system assistance for medical decision making (e.g. radiotherapy dose
planner).
26
Note: MDSW that is intended specifically to replace a part or component of a device and that significantly changes the
performance or safety characteristics or the intended purpose of the device shall be considered to be a device and shall meet
the requirements laid down in this Regulation (see Article 23.2 (2)).

Page 18 of 28
This raises the issue as to whether the whole product can be CE marked when not all applications have
a medical purpose.
Computer programmes used in healthcare can have applications which consist of both medical device
and non-medical device modules.
The modules which are subject to the Medical Devices Regulations (figure 1 and 2) must comply with
the requirements of the medical device regulations and must carry the CE marking. The non-medical
device modules are not subject to the requirements for medical devices. It is the obligation of the
manufacturer to identify the boundaries and the interfaces of the different modules.
The boundaries of the modules which are subject to the medical device regulations should be clearly
identified by the manufacturer based on the intended use.
If the modules which are subject to the medical device regulations are intended for use in combination
with other modules of the whole software structure, other devices or equipment, the whole
combination, including the connection system, must be safe and must not impair the specified
performances of the modules which are subject to the medical device regulations.
27
8. Consideration of changes to an MDSW
Manufacturers shall evaluate the potential impact of any changes to the function, intended use,
essential design, and manufacturing characteristics on the software’s qualification as MDSW and its
classification (including the classification of the combination of the MDSW with another medical
device).
It is to be noted that a change to or the addition of functionality to a software may lead it to be
qualified as MDSW, or a revision of the classification of the MDSW. Similarly, a module that is added
to a software might be qualified as a MDSW on its own.
When determining the risk class of a combination of a modified MDSW and a medical device, the
intended purpose and functionality of that (new) combination must be considered.
Note: For all MDSW, the manufacturer shall ensure safety and performance throughout the lifecycle
of the software, through a continuous process of clinical and/or performance evaluation and risk
management.
9. Annex I: Illustrative examples of qualification of software
used in the healthcare environment
Software for a medical purpose is rapidly evolving. Thus, the list of examples provided below is not
exhaustive. The examples have been drafted in light of today’s state of the art, in order to give the
reader a better understanding of the application of the principles set out in the guideline.
The Manual on borderline and classification in the Community regulatory framework for medical
devices contains many examples related to qualification of software and apps, under the current
27
i.e. general safety and performance requirements 14.1 of EU MDR 2017/745 and 13.1 of EU IVDR 2017/746

Page 19 of 28
Directives
28
. The Manual is currently under revision for adaptation to MDRs. In light of the
technological progress, further examples will be regularly published in both the Manual and in this
guidance.
a) Hospital Information Systems
Hospital Information Systems mean, in this context, systems that support the process of patient
management. Typically they are intended for patient admission, for scheduling patient appointments,
for insurance and billing purposes.
These Hospital Information Systems are not qualified as medical devices. However, they may be used
with additional modules, as described hereafter. These modules might be qualified in their own right
as medical devices.
b) Decision Support Software
In general, these are computer based tools which combine general medical information databases and
algorithms with patient-specific data. They are intended to provide healthcare professionals and/or
users with recommendations for diagnosis, prognosis, monitoring and treatment of individual patients.
Based on Figure 1, they are qualified as medical devices.
• Radiotherapy treatment planning systems
29
are intended to calculate the dosage of ionizing
irradiation to be applied to a specific patient. They are considered to control, monitor or
directly influence the source of ionizing radiation and are qualified as medical devices.
• Drug planning systems (e.g. chemotherapy) are intended to calculate the drug dosage to be
administered to a specific patient and therefore are qualified as medical devices.
• Computer Aided Detection systems are intended to provide information that may suggest or
exclude medical conditions are qualified as medical devices (MDSW). For example, such
systems would be able to automatically analyse x-ray images or interpret ECGs.
c) Information Systems
Information Systems that are intended only to transfer, store, convert, format, archive data are not
qualified as medical devices in themselves. However, they may be used with additional modules
which maybe qualified in their own right as medical devices (MDSW).
c.1.) Electronic Patient Record Systems
Electronic patient record systems are intended to store and transfer electronic patient records. They
archive all kinds of documents and data related to a specific patient. The electronic patient records
should not be qualified as a medical device, i.e. an electronic patient record that simply replaces a
patient’s paper file does not meet the definition of a medical device. The modules used with electronic
patient record system modules that might be qualified in their own right as medical devices (MDSW)
are for example:
• An image viewer with functionality for diagnosis based on digital images;
• A medication module
28
http://ec.europa.eu/DocsRoom/documents/12867/attachments/1/translations/en/renditions/native
29
See EN 62083 “Requirements for the safety and radiotherapy treatment planning systems”

Page 20 of 28
c.1.1.) Clinical Information Systems – CIS / Patient Data Management Systems – PDMS
A CIS/PDMS is a software-based system primarily intended to store and transfer patient information
generated in association with the patient’s intensive care treatment (e.g. intensive care units).
Usually the system contains information such as patient identification, vital intensive care parameters
and other documented clinical observations.
These CIS/PDMS are not qualified as medical devices.
Modules that are intended to provide additional information that contributes to diagnosis, therapy and
follow-up (e.g. generate alarms) are qualified as medical devices.
c.1.2.) Pre-hospital Electrocardiograph (ECG) System
A system for managing pre-hospital ECG is a software-based system intended for ambulance services
to store and transfer information from patients to a doctor at remote location. Usually the system
contains information about patient identification, vital parameters and other documented clinical
observations. These Pre-hospital Electrocardiograph (ECG) Systems are not qualified as medical
devices.
Modules that create and provide new patient treatment information to the paramedics or to the doctor
at a remote location to start the patient’s treatment while the patient is being transported are qualified
as medical devices.
c.1.3.) Picture Archive Communication System (PACS)
The Manual on Borderline and Classification in the Community Regulatory Framework for Medical
Devices addresses the qualification of PACS.
30
The transposition of this Directive Guidance to the
Regulations is currently underway and will be published at
https://ec.europa.eu/growth/sectors/medical-devices/new-regulations/guidance_en
d) Communication Systems
The healthcare sector uses communication systems (e.g. email systems, mobile telecommunication
systems, video communication systems, paging, etc.) to transfer electronic information. Different
types of messages are sent such as prescription, referrals, images, patient records, etc.
Most of the communication systems handle types of messages other than medical information. This
communication system is intended for general purposes, and is used for transferring both medical and
non-medical information.
Communication systems are normally based on software for general purposes, and do not fall within
the definition of a medical device.
Communication system modules might be used with other modules that might be qualified in their
own right as medical devices (MDSW).
A software module generating alarms based on the monitoring and analysis of patient specific
physiological parameters is qualified as a medical device (MDSW).
d.1) Telemedicine systems
Telemedicine Systems are intended to allow monitoring and/or delivery of healthcare to patients at
locations remote from where the healthcare professional is located.
30
https://ec.europa.eu/docsroom/documents/35582/attachments/1/translations/en/renditions/native
Page 21 of 28
d.1.1.) Telesurgery
Telesurgery is intended to conduct a surgical procedure from a remote location. Virtual reality
technology may be used to support a remote surgeon to control a surgical robot performing the
surgical procedure.
Telesurgery systems should be qualified as medical devices according to Figure 2 of this document.
Remote control software used in combination with telesurgery robots is a software that drives or
influences the use of a medical device. Communication modules themselves are not medical devices.
Other modules that are intended to influence the surgery procedure are qualified as medical devices
(MDSW).
e) Web systems for monitoring of data
A web system for the monitoring of clinical data typically interacts with a medical device (e.g.
implanted devices or homecare devices), and uses a transmitter to send the information over the
internet, a landline telephone or a mobile network.
The information is collected and stored on a web server usually run by an external party who is
generally the manufacturer of the system. The information can be reached by authorized health
professionals or the patient through an internet connection.
• Monitoring of performance of medical devices:
Modules that are intended to monitor the medical performance of medical devices fall under
the medical device regulations.
This includes the clinical performance and failures that could affect medical performance of
the device. One example of such a product is a web system for monitoring of active implants
such as pacemakers or Intra Cardiac Defibrillators (ICDs).
• Monitoring of non-medical performance of medical devices
Modules that are intended to perform administrative monitoring of non-medical performance
of medical devices do not necessarily fall under the scope of the medical devices regulations.
Software for the monitoring of medical devices in hospital systems for the purpose of
maintenance and repair.
f) In vitro diagnostic medical device software
f.1.) Laboratory Information Systems (LIS) and Work Area Managers (WAM)
Laboratory Information System (LIS) and Work Area Managers (WAM) mean, in this context,
systems that support the process from patient sample to patient result. Typically, they have pre-
analytical functions for ordering, sorting and distribution of test samples.
The main task is the management and validation of incoming information obtained from in vitro
diagnostic medical device analysers connected to the system, such as calibration, quality control,
product expiry and feedback (e.g. retesting of samples needed) through interconnections with various
analytical instruments (technical and clinical validation).
Finally the post-analytical process allows communication of laboratory results, statistics and optional
reporting to external databases.
The software normally supports the following functions:
• Ordering of laboratory tests, samples with labels and sorting;
• Technical and clinical validation, connection to analytic instruments;
• Laboratory results and reports on paper, fax or electronic records that can be directly returned
to e.g. the ordering clinic’s patient record;
• Analytical instruments can be interfaced with Hospital Information Systems (HIS), Electronic
Patient Record Systems, Infectious control databases, etc.
Page 22 of 28
Note: software intended to modify the representation of available in vitro diagnostic medical device
results is not considered an in vitro diagnostic medical device, e.g. basic operations of arithmetic (e.g.
mean, conversion of units) and/or plotting of results in function of time, and/or a comparison of the
result to the limits of acceptance set by the user.
The results are available, readable and understandable without the intervention of the software.
Laboratory Information Systems (LIS) and Work Area Managers (WAM) are not qualified as
medical devices in themselves. However, they may be used with additional modules. These modules
might be qualified in their own right as medical devices.
A module whose intended purpose is to assess the criticality of tests required and to perform
automatic reprioritisation of the order based on patient data is qualified as a MDSW.
f.2.) Expert system
MDSW which is intended to provide information within the scope of the in vitro diagnostic medical
devices definition by capturing and analysing together one or multiple results obtained for one patient
by means of in vitro examination of body samples (possibly combined with information from medical
devices and non-medical devices).
• MDSW that integrates genotype of multiple genes to predict risk a disease or medical
condition developing or recurring;
• MDSW that uses an algorithm to characterise viral resistances to various drugs, based on a
nucleotide sequence generated by genotyping assays. This software serves to generate new
information (virus resistance profile) from available information on the genotype of the virus;
• MDSW intended to be used in microbiology for the identification of clinical isolates and/or the
detection of antimicrobial resistances.
Refer to Figure 2 of this guidance if data is obtained from both in vitro diagnostic medical devices and
medical devices.
f.3.) Interpretation of raw data
In the case where MDSW is necessary to render raw data, readable for the user, obtained from an in
vitro diagnostic medical device by means of in vitro examination of body samples, this MDSW is to
be considered driving or influencing the use of the in vitro diagnostic medical device when it is
specifically intended to be used together with this in vitro diagnostic medical device to enable it to be
used in accordance with its intended purpose.
MDSW intended for the analysis and interpretation of enzyme-linked immunosorbent assay (ELISA)
reader optical density results, line patterns or spot patterns of a blot.
f.4.) Home care monitoring, wired or mobile
Software intended for archiving patient results or for transferring results obtained from in vitro
diagnostic medical devices from the home environment to the healthcare provider is not an in vitro
diagnostic medical device. The results are available, readable and understandable by the user without
the intervention of the software.
f.5.) Image Management System (IMS)
An IMS is a software-based system primarily intended to be networked with digital pathology
systems, e.g., whole slide scanners, scanning microscopes, as well as LIS. It does not contain controls
for the direct operation of the digital pathology systems, and is intended to access, display, annotate,
manage, store, archive and share collections of digitised patient images. IMS may be configured to
Page 23 of 28
provide limited or extensive capabilities to further visualise or analyse patient images acquired from
networked digital pathology systems.
An IMS only used for viewing, archiving and transmitting images are not considered medical devices
in themselves. However, these IMS may be used with additional modules that might be qualified in
their own right as medical devices (MDSW).
IMS that incorporate functions to support post-processing of images for diagnostics purposes, e.g.,
image processing functions which alter image data or complex quantitative functions to aid in
diagnosis, are qualified as MDSW.

Page 24 of 28
10. Annex II - Qualification examples of Medical Device
Software (MDSW) according to Figures 1 and 2
Figure 1 - Example 1:
A software module which runs on an in vitro diagnostic medical device instrument and tracks how the
laboratory is performing in real-time on key operational metrics such as test volumes, turnaround
times, pending tests, and quality control. Its intent is to improve a laboratory’s operations by providing
real-time monitoring of performance metrics that can drive change management and continuous
improvement initiatives within the lab. The software is configurable so that customers can choose the
metrics on which they would like to focus.
Qualification: Step 1 is concluded with a “yes” as the software is a product which uses a set of
instructions (or algorithm) to process input data and create output data. Step 2 determines that the
software is not an MDR Annex XVI device, nor is it an accessory for a medical device, nor a software
driving or influencing the use of a medical device. Step 3 is answered “yes” since the software is
doing more than storage, archival, communication or simple search of information. Step 4 is answered
“no” as the software does not perform this action for the benefit of individual patients. The conclusion
is that the software does not fall under the Medical Devices Regulations. This is appropriate since the
software is intended to be a Laboratory Information Systems (LIS), which is not considered a medical
device.
Figure 2 - Example 2:
MDSW intended to generate a risk score in order to trigger care processes to help reduce ICU
transfers, readmissions, adverse events and length of stay. The risk score by default includes
respiratory rate, heart rate, blood pressure and SpO2, but a user can configure it to include other
parameters, including in vitro diagnostic medical device results.
Qualification: In decision Step 1, the MDSW can be understood to meet criteria (a), (f) and (h), it is
therefore answered “yes”. Step 2 is answered with a “No” as an in vitro diagnostic medical device
result may be included in the in the calculation. Step 3 directs the significance of the medical device
derived information as driving the intended purpose, resulting in the qualification of the software as an
MD MDSW (as the data received from the in vitro diagnostic medical device is not deemed decisive
for the overall calculation result (output) achieved by the MDSW)
Figure 2 - Example 3:
A MDSW algorithm intended to provide information on the statistical predisposition for Down
syndrome (Trisomy 21) and Edwards syndrome (Trisomy 18) in the first and second trimesters of
pregnancy.
31
The MDSW analyses input data from various in vitro diagnostic medical device assays
32
as well as, ultrasound measurements of the nasal bone or neck fold.
The MDSW provides
clinicians/obstetricians with a risk factor score for a foetus’s likelihood of having genetic mutations in
the first or second trimester of pregnancy. The risk score suggests whether or not additional
diagnostic testing is needed to confirm the genetic mutations of Trisomy 21, Trisomy 18.
33
Qualification: Step 1 can be answered “yes” as the software bears a medical purpose and fulfils the
definition of MDSW. The MDSW meets criteria (c) as it provides information according to the in vitro
31
The software can also detect neural tube defects (NTD) in the second trimester as well as the risk for Patau’s syndrome
(Trisomy 13).
32
AFP (LKAP, L2KAP), Unconjugated Estriol (LKUE3, L2KUE3), hCG (LKCG, L2KCG), free β-hCG (LKFB, L2KFB),
PAPP A (LKPC, L2KPC)
33
or neural tube defects and Trisomy 13
Page 25 of 28
diagnostic medical devices definition. Decision step 2 is answered “no” as an imaging measurement is
included in the calculation. Step 3 is answered “yes” as the intended purpose is substantially driven by
in vitro diagnostic medical device data resulting in the qualification of the software as an IVD MDSW
(as the data received from the in vitro diagnostic medical devices (markers) are deemed decisive for
the overall calculation result (output) achieved by the MDSW).
Figure 2- Example 4:
A bioinformatics MDSW intended to analyse digital Next Generation Sequencing (NGS) raw data
coming from sequenced patient’s cancer genomes. It allows the detection and visualisation of somatic
genome alterations (such as substitutions, small insertions and deletions (indels), copy number
alterations, and genomic rearrangements) across a selected number of genes. Additionally, it is also
capable of determining genomic signatures* (such as microsatellite instability [MSI] and/or tumour
mutational burden [TMB]). The types of somatic genome alterations and genomic signatures detected
depend on the test chosen. The MDSW assists the user in identifying and visualising genomic
alterations and is intended to identify somatic genome alterations to support diagnosis and treatment
decisions.
Qualification: Decision step 1 is concluded with a “yes” as the MDSW is intended for analysing
congenital data to provide information on the predisposition to a medical condition or disease, thus
meeting criteria (b) and (c) laid out in the decision step. As the MDSW processes data coming only
from in vitro diagnostic medical devices into the calculation, then the software is qualified as an IVD
MDSW according to Step 2.

Page 26 of 28
11. Annex III - Usability of the IMDRF risk classification
framework in the context of the MDR
34
The table below, which is intended for illustrative purposes only, may provide operators placing
MDSW on the EU market with some useful indicative orientation on the risk class applicable to their
products as a result of the application of Rule 11 a of the MDR.
Note: MDR 2017/745 Sub-rule 11(a), point i., referring above to MDR class III, aligns with IMDRF
risk category IV. Sub-rule 11(a), point ii.,, referring above to MDR class IIb, aligns with IMDRF risk
category III products mentioned in section 7.2 of the mentioned IMDRF document. The IMDRF risk
category II and IMDRF risk category I products are classified as MDR class IIa as per Rule 11.
This table does not take into account MDSW which is Class I.
Significance of Information provided by the
MDSW to a healthcare situation related to
diagnosis/therapy
State of Healthcare
situation or patient condition
High
Treat or
diagnose
~ IMDRF 5.1.1
Medium
Drives clinical
management
~ IMDRF 5.1.2
Low
Informs clinical
management
(everything else)
Critical situation
or patient
condition
~ IMDRF 5.2.1
Class III
Category IV.i
Class IIb
Category III.i
Class IIa
Category II.i
Serious situation
or patient
condition
~ IMDRF 5.2.2
Class IIb
Category III.ii
Class IIa
Category II.ii
Class IIa
Category I.ii
Non-serious
situation or
patient condition
(everything else)
Class IIa
Category II.iii
Class IIa
Category I.iii
Class IIa
Category I.i
Table 1: Classification Guidance on Rule 11
34
Annex III and IV are not relevant for IVD MDSW. See section 4.2 in order to classify IVD MDSW. MDCG Guidance on
Classification of IVDs should also be considered.
Page 27 of 28
12. Annex IV – Classification examples
The examples are provided for guidance purposes only, to illustrate how a particular rule may be
applied to a device. The indicated classification in the example is not a confirmation of the final
classification of the device, as other rules might also be considered.
Moreover, the proposed classification reflects the specific intended purpose, or the healthcare context
or situation, in which the device is used as described in the example itself. Any change to the intended
purpose or the healthcare context/situation in which that same device is used might result in a different
risk class.
• MDSW intended to perform diagnosis by means of image analysis for making treatment
decisions in patients with acute stroke should be classified as class III under Rule 11(a)
- IMDRF Risk Category IV.i as the healthcare situation (stroke) is critical and the
significance of the information is “treat or diagnose”.
• Cognitive therapy MDSW that includes a diagnostic function which is intended to feed back
to the software to determine follow-up therapy, e.g. software adapts treatment of depression
based on diagnostic feedback, should be in class III per Rule 22. When a specialist
determines the necessary cognitive therapy based on the outcome provided by the MDSW, the
MDSW would be classified as class IIa per Rule 11(a).
- IMDRF Risk Category II.ii as the healthcare situation is serious and the significance
of the information is to “drive clinical management”.
• Medical devices including MDSW intended to be used for continuous surveillance of vital
physiological processes in anaesthesia, intensive care or emergency care should be classified
as class IIb per (Rule 11(b)).
• Medical devices, including MDSW intended to monitor physiological processes that are not
considered to be vital, and devices intended to be used to obtain readings of vital
physiological signals in routine check-ups including monitoring at home should be classified
as class IIa (Rule 11(b)).
• A mobile app intended to analyse a user’s heartbeat, detect abnormalities and inform a
physician accordingly should be classified as class IIb per Rule 11(a), if the information
provided by the software is intended to guide the physician in the diagnosis.
- IMDRF Risk Category III.i as the information drives clinical management.
• Diagnostic MDSW intended for scoring depression based on inputted data on a patient’s
symptoms (e.g. mood, anxiety) should be classified as class IIb under Rule 11(a),
- (IMDRF Risk Category III.ii) as the healthcare situation (depression) is serious and
the significance of the information is “diagnosis”).
• Ambulatory respiratory ventilation systems MDSW intended for long-term use (e.g. at home)
that alert the user/operator to any disconnection or deviation to the programmed respiratory
volume should be classified as class IIb per Rule 9.
• Active devices, such as electronic thermometers and stethoscopes, which include MDSW
intended for direct diagnosis may be classified as class IIa per Rule 10, third indent since
body temperature and heart rate are considered decisive information for diagnosis
(implementing rule 3.7), where the nature of the variations of these parameters would not
result in immediate danger to the patient.
• MDSW intended to rank therapeutic suggestions for a health care professional based on
patient history, imaging test results, and patient characteristics, for example, MDSW that lists
and ranks all available chemotherapy options for BRCA-positive individuals, should be
classified as class IIa per Rule 11(a)
Page 28 of 28
- IMDRF Risk Category II.i as it informs clinical management for cancer, a critical
disease.
• MDSW app intended to support conception by calculating the user’s fertility status based on a
validated statistical algorithm. The user inputs health data including basal body temperature
(BBT) and menstruation days to track and predict ovulation. The fertility status of the current
day is reflected by one of three indicator lights: red (fertile), green (infertile) or yellow
(learning phase/cycle fluctuation). This MDSW app should be classified as class I per Rule
11c.
